Establishment of prognostic model for severe primary graft dysfunction in patients with idiopathic pulmonary fibrosis after lung transplantation
-
摘要:
目的 探索性构建基于机器学习算法预测特发性肺纤维化(IPF)肺移植受者术后原发性移植物功能障碍(PGD)的预后模型。 方法 回顾性分析226例行肺移植手术的IPF患者的资料。所有入组患者按7∶3随机划分为训练集和测试集。利用正则化logistic回归、随机森林、支持向量机和人工神经网络4种方法,通过变量筛选、构建模型、模型调优流程构建模型。使用受试者工作特征曲线下面积(AUC)、阳性预测值、阴性预测值和准确度进行模型性能评估。 结果 共筛选出16个关键特征用于建模。4种预后模型的AUC值均>0.7。DeLong检验和McNemar检验发现模型间性能差异无统计学意义(均为P>0.05)。 结论 基于4种机器学习算法初步构建了肺移植术后3级PGD的预后模型。各模型整体预测性能相似,均可对IPF患者肺移植术后3级PGD进行较好的预测。 -
关键词:
- 肺移植 /
- 特发性肺纤维化 /
- 机器学习 /
- 原发性移植物功能障碍 /
- 随机森林 /
- logistic回归 /
- 支持向量机 /
- 人工神经网络
Abstract:Objective To explore the establishment of a prognostic model based on machine learning algorithm to predict primary graft dysfunction (PGD) in patients with idiopathic pulmonary fibrosis (IPF) after lung transplantation. Methods Clinical data of 226 IPF patients who underwent lung transplantation were retrospectively analyzed. All patients were randomly divided into the training and test sets at a ratio of 7:3. Using regularized logistic regression, random forest, support vector machine and artificial neural network, the prognostic model was established through variable screening, model establishment and model optimization. The performance of this prognostic model was assessed by the area under the receiver operating characteristic curve (AUC), positive predictive value, negative predictive value and accuracy. Results Sixteen key features were selected for model establishment. The AUC of the four prognostic models all exceeded 0.7. DeLong and McNemar tests found no significant difference in the performance among different models (both P>0.05). Conclusions Based on four machine learning algorithms, the prognostic model for grade 3 PGD after lung transplantation is preliminarily established. The overall prediction performance of each model is similar, which may predict the risk of grade 3 PGD in IPF patients after lung transplantation. -
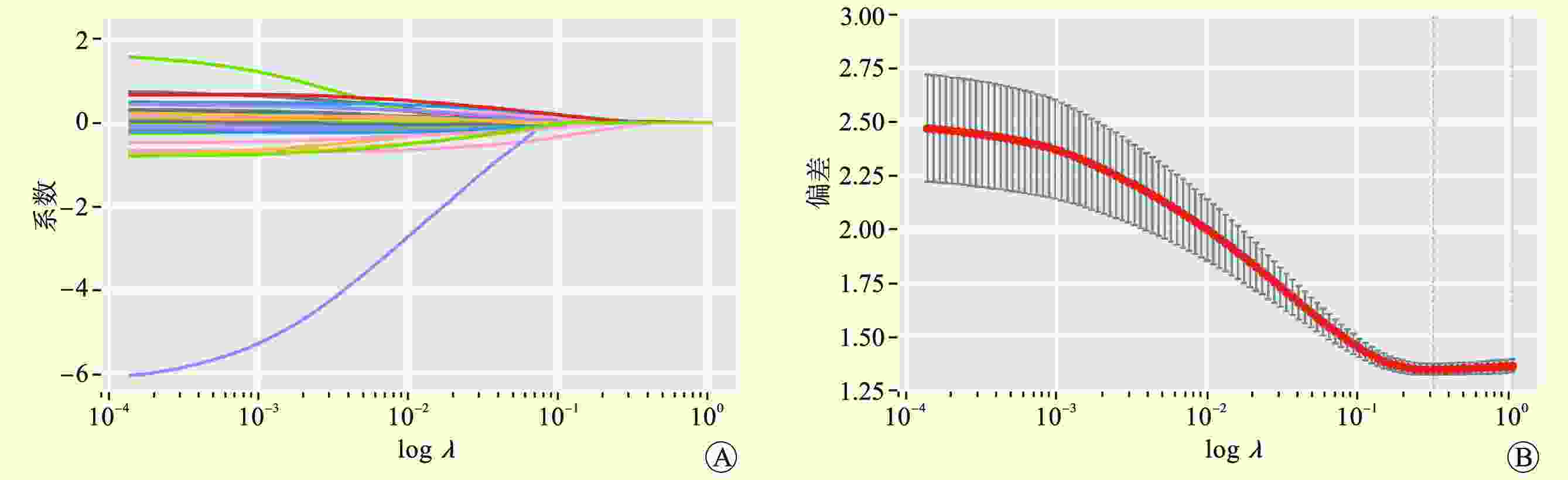
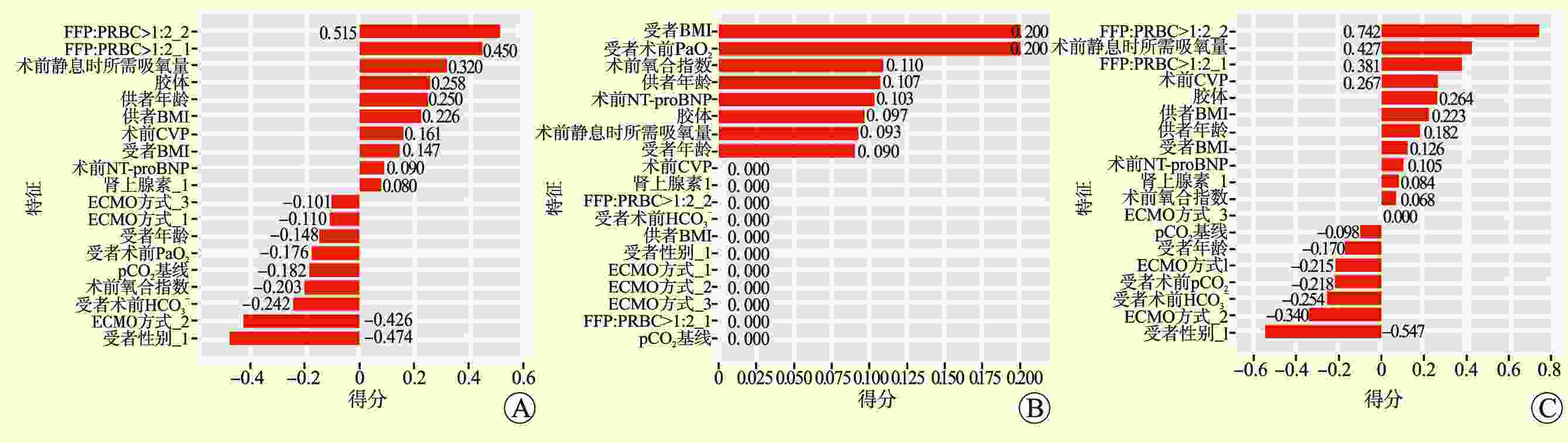
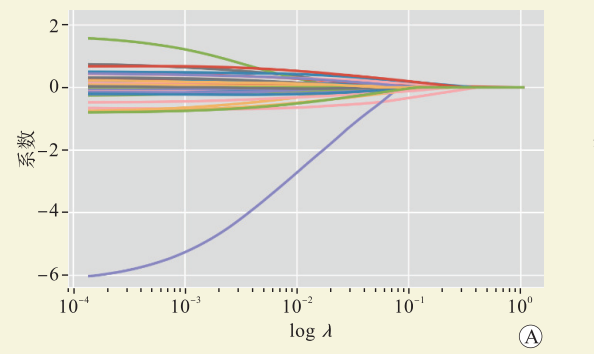
表 1 弹性网络模型系数
Table 1. Coefficient of elastic network model
变量 系数 供者变量 BMI 0.015 961 年龄 0.006 710 受者变量 性别 −0.067 410 BMI 0.009 635 年龄 −0.004 860 术前静息时所需吸氧量 0.024 346 术前PaO2 −0.001 940 pCO2基线 −0.001 710 术前氧合指数 −0.001 130 术前NT-proBNP 0.000 031 术前CVP 0.007 643 术前${\mathrm{HCO}}_3^{\,\,- }$ −0.005 980 移植变量 FFP∶PRBC>1∶2 0.024 577 ECMO方式 0.009 942 肾上腺素 0.000 225 胶体 0.000 103 表 2 各模型测试集评估参数
Table 2. Evaluation parameters for each model test set
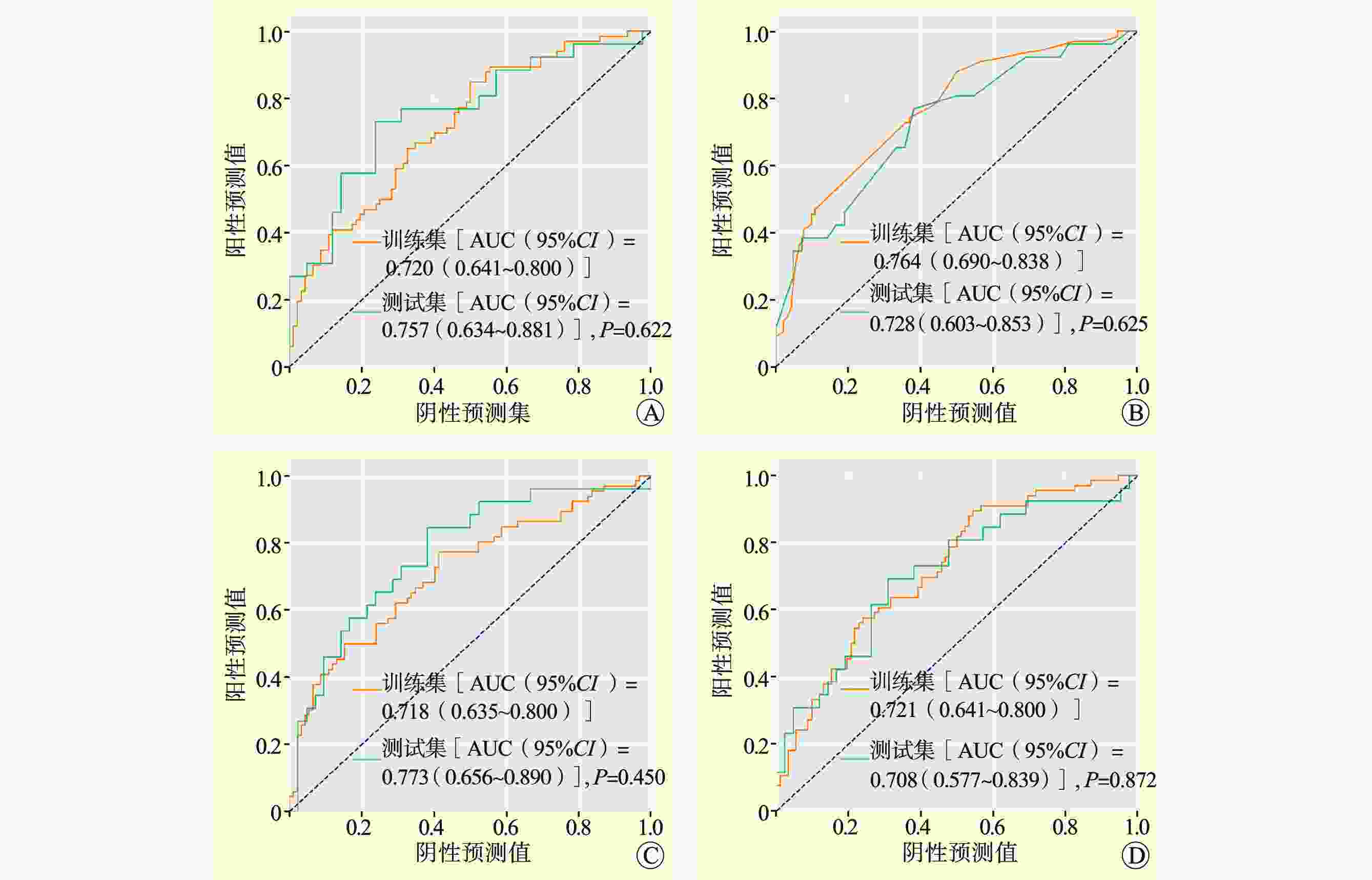
模型 AUC
(95%CI①)灵敏度
(95%CI)特异度
(95%CI)PPV
(95%CI)NPV
(95%CI)准确度
(95%CI)LR 0.76
(0.63~0.88)0.89
(0.76~1.01)0.43
(0.28~0.58)0.49
(0.35~0.63)0.86
(0.71~1.01)0.60
(0.49~0.72)RF 0.73
(0.60~0.85)0.81
(0.66~0.96)0.50
(0.35~0.65)0.50
(0.35~0.65)0.81
(0.66~0.96)0.62
(0.50~0.73)SVM 0.77
(0.66~0.89)0.85
(0.71~0.99)0.50
(0.35~0.65)0.51
(0.36~0.66)0.84
(0.70~0.98)0.63
(0.52~0.75)ANN 0.71
(0.58~0.84)0.85
(0.71~0.99)0.38
(0.23~0.53)0.46
(0.32~0.60)0.80
(0.63~0.98)0.56
(0.44~0.68)注:①CI为可信区间。 表 3 各模型ROC曲线DeLong检验结果
Table 3. DeLong test results of ROC curves for each model
模型 LR RF SVW ANN LR −① Z=0.585, P=0.559 Z=−0.449, P=0.618 Z=1.326, P=0.185 RF − − Z=−0.850, P=0.395 Z=−0.375, P=0.708 SVW − − − Z=−1.516, P=0.129 ANN − − − − 注:①−为无数据。 表 4 McNemar检验结果
Table 4. McNemar test results
模型 LR RF SVW ANN LR −① χ2=0.000, P=1.000 χ2=0.125, P=0.724 χ2=0.364, P=0.546 RF − − χ2=0.000, P=1.000 χ2=0.409, P=0.522 SVW − − − χ2=1.455, P=0.228 ANN − − − − 注:①−为无数据。 -
[1] DIAMOND JM, LEE JC, KAWUT SM, et al. Clinical risk factors for primary graft dysfunction after lung transplantation[J]. Am J Respir Crit Care Med, 2013, 187(5): 527-534. DOI: 10.1164/rccm.201210-1865OC. [2] CLAUSEN E, CANTU E. Primary graft dysfunction: what we know[J]. J Thorac Dis, 2021, 13(11): 6618-6627. DOI: 10.21037/jtd-2021-18. [3] M LAMBERT E, A WUYTS W, YSERBYT J, et al. Statins: cause of fibrosis or the opposite? effect of cardiovascular drugs in idiopathic pulmonary fibrosis[J]. Respir Med, 2021, 176: 106259. DOI: 10.1016/j.rmed.2020.106259. [4] GAIKWAD AV, LU W, DEY S, et al. Endothelial-to-mesenchymal transition: a precursor to pulmonary arterial remodelling in patients with idiopathic pulmonary fibrosis[J]. ERJ Open Res, 2023, 9(2): 00487-2022. DOI: 10.1183/23120541.00487-2022. [5] BALESTRO E, COCCONCELLI E, TINÈ M, et al. Idiopathic pulmonary fibrosis and lung transplantation: when it is feasible[J]. Medicina (Kaunas), 2019, 55(10): 702. DOI: 10.3390/medicina55100702. [6] GUO H, SUN J, ZHANG S, et al. Progress in understanding and treating idiopathic pulmonary fibrosis: recent insights and emerging therapies[J]. Front Pharmacol, 2023, 14: 1205948. DOI: 10.3389/fphar.2023.1205948. [7] 许红阳, 王大鹏, 姜淑云, 等. 肺移植治疗良性终末期肺病受者术后1年内生存的危险因素分析[J]. 中华危重病急救医学, 2021, 33(7): 832-837. DOI: 10.3760/cma.j.cn121430-20200729-00550.XU HY, WANG DP, JIANG SY, et al. Risk factors analysis for 1-year postoperative survival of patients with benign end-stage lung diseases after lung transplantation[J]. Chin Crit Care Med, 2021, 33(7): 832-837. DOI: 10.3760/cma.j.cn121430-20200729-00550. [8] STEYERBERG EW, MOONS KG, VAN DER WINDT DA, et al. Prognosis Research Strategy (PROGRESS) 3: prognostic model research[J]. PLoS Med, 2013, 10(2): e1001381. DOI: 10.1371/journal.pmed.1001381. [9] PÉREZ-TERÁN P, ROCA O, RODRÍGUEZ-PALOMARES J, et al. Prospective validation of right ventricular role in primary graft dysfunction after lung transplantation[J]. Eur Respir J, 2016, 48(6): 1732-1742. DOI: 10.1183/13993003.02136-2015. [10] DIAMOND JM, LEDERER DJ, KAWUT SM, et al. Elevated plasma long pentraxin-3 levels and primary graft dysfunction after lung transplantation for idiopathic pulmonary fibrosis[J]. Am J Transplant, 2011, 11(11): 2517-2522. DOI: 10.1111/j.1600-6143.2011.03702.x. [11] GIRGIS RE, HADLEY RJ, MURPHY ET. Pulmonary, circulatory and renal considerations in the early postoperative management of the lung transplant recipient[J]. Glob Cardiol Sci Pract, 2023, 2023(3): e202318. DOI: 10.21542/gcsp.2023.18. [12] COLLINS GS, REITSMA JB, ALTMAN DG, et al. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement[J]. BMJ, 2015, 350: g7594. DOI: 10.1136/bmj.g7594. [13] SGALLA G, KULKARNI T, ANTIN-OZERKIS D, et al. Update in pulmonary fibrosis 2018[J]. Am J Respir Crit Care Med, 2019, 200(3): 292-300. DOI: 10.1164/rccm.201903-0542UP. [14] CHRISTIE JD, CARBY M, BAG R, et al. Report of the ISHLT Working Group on primary lung graft dysfunction part II: definition. a consensus statement of the International Society for Heart and Lung Transplantation[J]. J Heart Lung Transplant, 2005, 24(10): 1454-1459. DOI: 10.1016/j.healun.2004.11.049. [15] SNELL GI, YUSEN RD, WEILL D, et al. Report of the ISHLT Working Group on primary lung graft dysfunction, part I: definition and grading-a 2016 consensus group statement of the International Society for Heart and Lung Transplantation[J]. J Heart Lung Transplant, 2017, 36(10): 1097-1103. DOI: 10.1016/j.healun.2017.07.021. [16] 邓猛聪, 王昕, 吴尚纯, 等. 受试者工作特征曲线(ROC曲线)的应用分析[J]. 中国计划生育学杂志, 2024, 32(2): 467-473. DOI: 10.3969/j.issn.1004-8189.2024.02.045.DENG MC, WANG X, WU SC, et al. Application analysis of receiver operator characteristic curve[J]. Chin J Family Plan, 2024, 32(2): 467-473. DOI: 10.3969/j.issn.1004-8189.2024.02.045. [17] ZHANG KC, NARANG N, JASSERON C, et al. Development and validation of a risk score predicting death without transplant in adult heart transplant candidates[J]. JAMA, 2024, 331(6): 500-509. DOI: 10.1001/jama.2023.27029. [18] PELOSO A, NAESENS M, THAUNAT O. The dawn of a new era in kidney transplantation: promises and limitations of artificial intelligence for precision diagnostics[J]. Transpl Int, 2023, 36: 12010. DOI: 10.3389/ti.2023.12010. [19] CHEN C, CHEN B, YANG J, et al. Development and validation of a practical machine learning model to predict sepsis after liver transplantation[J]. Ann Med, 2023, 55(1): 624-633. DOI: 10.1080/07853890.2023.2179104. [20] BHAT M, RABINDRANATH M, CHARA BS, et al. Artificial intelligence, machine learning, and deep learning in liver transplantation[J]. J Hepatol, 2023, 78(6): 1216-1233. DOI: 10.1016/j.jhep.2023.01.006. [21] ATAGUN GUNEY P, UYGUN KIZMAZ Y. Bronchial culture growth from the donor and recipient as predictive factors in the detection of primary graft dysfunction and pneumonia after lung transplant[J]. Exp Clin Transplant, 2022, 20(10): 930-936. DOI: 10.6002/ect.2021.0496. [22] JENKINS JA, VERDINER R, OMAR A, et al. Donor and recipient risk factors for the development of primary graft dysfunction following lung transplantation[J]. Front Immunol, 2024, 15: 1341675. DOI: 10.3389/fimmu.2024.1341675. [23] MOON S, PARK MS, LEE JG, et al. Risk factors and outcome of primary graft dysfunction after lung transplantation in Korea[J]. J Thorac Dis, 2016, 8(11): 3275-3282. DOI: 10.21037/jtd.2016.11.48. [24] LIU Y, LIU Y, SU L, et al. Recipient-related clinical risk factors for primary graft dysfunction after lung transplantation: a systematic review and meta-analysis[J]. PLoS One, 2014, 9(3): e92773. DOI: 10.1371/journal.pone.0092773. [25] LOOR G, HUDDLESTON S, HARTWIG M, et al. Effect of mode of intraoperative support on primary graft dysfunction after lung transplant[J]. J Thorac Cardiovasc Surg, 2022, 164(5): 1351-1361. DOI: 10.1016/j.jtcvs.2021.10.076. [26] FESSLER J, VALLÉE A, GUIRIMAND A, et al. Blood lactate during double-lung transplantation: a predictor of grade-3 primary graft dysfunction[J]. J Cardiothorac Vasc Anesth, 2022, 36(3): 794-804. DOI: 10.1053/j.jvca.2021.10.043. [27] EHRSAM JP, SCHUURMANS MM, LAAGER M, et al. Recipient comorbidities for prediction of primary graft dysfunction, chronic allograft dysfunction and survival after lung transplantation[J]. Transpl Int, 2022, 35: 10451. DOI: 10.3389/ti.2022.10451. [28] PORTEOUS MK, LEE JC, LEDERER DJ, et al. Clinical risk factors and prognostic model for primary graft dysfunction after lung transplantation in patients with pulmonary hypertension[J]. Ann Am Thorac Soc, 2017, 14(10): 1514-1522. DOI: 10.1513/AnnalsATS.201610-810OC. [29] 胡春兰, 刘民强, 于慧智, 等. 特发性肺纤维化合并肺动脉高压肺移植患者术后早期死亡危险因素分析[J]. 中华危重病急救医学, 2023, 35(2): 124-129. DOI: 10.3760/cma.j.cn121430-20220523-00506.HU CL, LIU MQ, YU HZ, et al. Risk factors of early death after lung transplantation in patients with idiopathic pulmonary fibrosis complicated with pulmonary arterial hypertension[J]. Chin Crit Care Med, 2023, 35(2): 124-129. DOI: 10.3760/cma.j.cn121430-20220523-00506. [30] IUS F, TUDORACHE I, WARNECKE G. Extracorporeal support, during and after lung transplantation: the history of an idea[J]. J Thorac Dis, 2018, 10(8): 5131-5148. DOI: 10.21037/jtd.2018.07.43. [31] FACCIOLI E, TERZI S, PANGONI A, et al. Extracorporeal membrane oxygenation in lung transplantation: indications, techniques and results[J]. World J Transplant, 2021, 11(7): 290-302. DOI: 10.5500/wjt.v11.i7.290. [32] WARRIOR K, SAYAD K, O'HARA CP, et al. Impact of acute exacerbation of idiopathic pulmonary fibrosis on lung transplant outcomes[J]. Transplantation, 2024,108(6):1460-1465.DOI: 10.1097/TP.0000000000004910. [33] LASKEY D, HOUSMAN B, DAWODU G, et al. Intraoperative extracorporeal support during lung transplantation: not just for the high-risk patient[J]. J Clin Med, 2023, 13(1): 192. DOI: 10.3390/jcm13010192. [34] HALPERN SE, WRIGHT MC, MADSEN G, et al. Textbook outcome in lung transplantation: planned venoarterial extracorporeal membrane oxygenation versus off-pump support for patients without pulmonary hypertension[J]. J Heart Lung Transplant, 2022, 41(11): 1628-1637. DOI: 10.1016/j.healun.2022.07.015. [35] TOYODA T, CERIER EJ, MANERIKAR AJ, et al. Recipient, donor, and surgical factors leading to primary graft dysfunction after lung transplant[J]. J Thorac Dis, 2023, 15(2): 399-409. DOI: 10.21037/jtd-22-974. [36] WORKU ET, WIN AM, PARMAR D, et al. Haematological trends and transfusion during adult extracorporeal membrane oxygenation: a single centre study[J]. J Clin Med, 2023, 12(7): 2629. DOI: 10.3390/jcm12072629. [37] YU Y, LIAN Z. Update on transfusion-related acute lung injury: an overview of its pathogenesis and management[J]. Front Immunol, 2023, 14: 1175387. DOI: 10.3389/fimmu.2023.1175387. [38] VAN WONDEREN SF, PETERS AL, GREY S, et al. Standardized reporting of pulmonary transfusion complications: development of a model reporting form and flowchart[J]. Transfusion, 2023, 63(6): 1161-1171. DOI: 10.1111/trf.17346. [39] SEAY T, GUINN N, MAISONAVE Y, et al. The association of increased FFP: RBC transfusion ratio to primary graft dysfunction in bleeding lung transplantation patients[J]. J Cardiothorac Vasc Anesth, 2020, 34(11): 3024-3032. DOI: 10.1053/j.jvca.2020.05.043. [40] DUAN Q, ZHANG Y, YANG D. Perioperative fluid management for lung transplantation is challenging[J]. Heliyon, 2023, 9(4): e14704. DOI: 10.1016/j.heliyon.2023.e14704. [41] BERGER G, KLORIN G, ISMAEL-BADARNEH R, et al. The cellular mechanisms of lung edema clearance: does the alveolar epithelium play a role?[J]. Harefuah, 2017, 156(10): 663-665. [42] MATHIS S, PUTZER G, SCHNEEBERGER S, et al. The endothelial glycocalyx and organ preservation-from physiology to possible clinical implications for solid organ transplantation[J]. Int J Mol Sci, 2021, 22(8): 4019. DOI: 10.3390/ijms22084019. [43] WANG G, ZHANG H, LIU D, et al. Resuscitation fluids as drugs: targeting the endothelial glycocalyx[J]. Chin Med J (Engl), 2022, 135(2): 137-144. DOI: 10.1097/CM9.0000000000001869. [44] AKBILGIC O, DAVIS RL. The promise of machine learning: when will it be delivered?[J]. J Card Fail, 2019, 25(6): 484-485. DOI: 10.1016/j.cardfail.2019.04.006. -




 下载:
下载: