Clinical analysis of the influence of gender on blood concentration of mycophenolic acid in recipients undergoing renal transplantation
-
摘要:
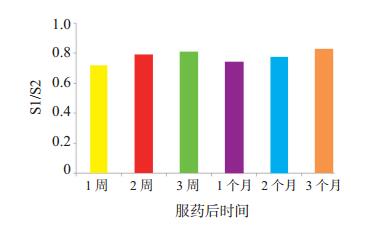
目的 探讨性别对肾移植术后受体霉酚酸(MPA)血药浓度的影响。 方法 以115例接受活体肾移植的受体作为研究对象, 根据性别将受体分为两组, 男性为S1组(61例), 女性为S2组(54例)。术后采用他克莫司(每次2 mg, 每日2次)+吗替麦考酚酯(MMF, 每次0.75 g, 每日2次)+泼尼松三联免疫抑制方案至少1周, 检测两组受体服药后1、2、3周和1、2、3个月的MPA血药谷浓度。分析两组受体不良反应发生情况。 结果 服药后1、2、3周和1、2、3个月, S1组受体的MPA血药谷浓度均低于S2组, 差异均有统计学意义(均为P < 0.05)。服药后不同时间点S1组与S2组受体MPA血药谷浓度均数的比值较稳定, 波动在0.71~0.84。S1组MPA血药谷浓度 < 1.0 μg/mL者的例数较多, 尤其是服药后1、2周, 达到44%、20%, 而S2组MPA血药谷浓度 > 3.5 μg/mL者的例数较多, 波动在30%~78%。S1组中4例发生急性排斥反应, 其中3例因急性排斥反应导致移植物失功; S2组中7例发生胃肠道紊乱, 3例发生肺部感染。 结论 性别对于肾移植受体术后服用MPA类药物的血药浓度有一定影响, 女性受体术后MPA血药谷浓度明显高于男性受体。 Abstract:Objective To investigate the influence of gender on the blood concentration of mycophenolic acid (MPA) in the recipients after renal transplantation. Methods A total of 115 recipients receiving living donor renal transplantation were recruited and divided into the S1 (61 male cases) and S2 groups (54 female cases).After the operation, triple immunosuppressive regimen of tacrolimus (2 mg for each time, twice daily) + mycophenolate mofetil (MMF, 0.75 g for each time, twice daily) + prednisone was administered for at least one week.The trough blood concentration of MPA was statistically compared between two groups at 1-, 2-, 3-week, 1-, 2- and 3-month after drug administration, and the incidence of adverse reactions were analyzed between two groups. Results At 1-, 2-, 3-week, 1-, 2- and 3-month after drug administration, the trough blood concentration of MPA in the S1 group was significantly lower than that in the S2 group (all P < 0.05).At different time points after drug administration, the ratio of the mean trough blood concentration of MPA between the S1 and S2 groups was relatively stable and fluctuated between 0.71 and 0.84.The percentage of recipients with trough blood concentration of MPA < 1.0 μg/mL in the S1 group was high, especially at 1 week (44%) or 2 week (20%) after drug administration.In the S2 group, the proportion of recipients with trough blood concentration of MPA > 3.5 μg/mL was high with the fluctuations ranging from 30% to 78%.In the S1 group, 4 cases suffered from acute rejection, in whom 3 patients had graft failure due to acute rejection.In the S2 group, 7 patients presented with gastrointestinal disorder and 3 had pulmonary infection. Conclusions Gender exerts certain influence on the trough blood concentration of MPA in the recipients after renal transplantation.The trough blood concentration of MPA in the female recipients is significantly higher than that in the male recipients. -
Key words:
- Gender /
- Estrogen /
- Renal transplantation /
- Mycophenolic acid /
- Mycophenolate mofetil /
- Blood concentration /
- UDP-glucuronosyltransferases /
- Genitein
-
表 1 两组受体服药后不同时间MPA血药谷浓度的比较
Table 1. Comparison of MPA trough blood concentration of recipients at different time after drug administration between two groups(x±s, μg/mL)
组别 n 服药后时间 1周 2周 3周 1个月 2个月 3个月 S1组 61 1.8±1.7 2.2±1.5 2.9±1.5 3.1±1.7 3.4±1.8 3.8±1.9 S2组 54 2.5±1.9 2.8±1.5 3.6±1.8 4.2±2.0 4.4±2.1 4.6±1.6 P值 < 0.05 < 0.05 < 0.05 < 0.05 < 0.05 < 0.05 表 2 两组受体服药后不同时间MPA血药谷浓度<1.0 μg/mL与>3.5 μg/mL的例数分布
Table 2. The case distribution of MPA trough blood concentration of recipients less than 1.0 μg/mL and greater than 3.5 μg/mL at different time after drug administration in the two groups[n(%)]
MPA血药谷浓度 S1组(n=61) S2组(n=54) 服药后1周 < 1.0 μg/mL 27(44) 15(28) > 3.5 μg/mL 10(16) 16(30) 服药后2周 < 1.0 μg/mL 12(20) 10(19) > 3.5 μg/mL 8(13) 20(37) 服药后3周 < 1.0 μg/mL 4(7)0 1(2)0 > 3.5 μg/mL 20(33) 24(44) 服药后1个月 < 1.0 μg/mL 2(3)0 0 > 3.5 μg/mL 19(31) 26(48) 服药后2个月 < 1.0 μg/mL 3(5)0 0 > 3.5 μg/mL 27(44) 32(59) 服药后3个月 < 1.0 μg/mL 2(3)0 1(2)0 > 3.5 μg/mL 32(52) 42(78) 表 3 两组受体服药后不良反应的发生情况
Table 3. Occurrence of adverse reactions in recipients after drug administration between two groups [n (%)]
组别 n 不良反应 移植物失功 急性排斥反应 感染 胃肠道紊乱 其他 S1组 61 3(5) 4(7) 1(2) 3(5)0 1(2) S2组 54 1(2) 1(2) 3(6) 7(13) 0 -
[1] THURET R, KLEINCLAUSS F, TERRIER N, et al.Challenges in renal transplantation[J].Prog Urol, 2016, 26(15):1001-1044.DOI: 10.1016/j.purol.2016.09.056. [2] FETURI FG, WEINSTOCK M, ZHAO W, et al.Mycophenolic acid for topical immunosuppression in vascularized composite allotransplantation:optimizing formulation and preliminary evaluation of bioavailability and pharmacokinetics[J].Front Surg, 2018, 5:20.DOI: 10.3389/fsurg.2018.00020. [3] SLOVAK JE, RIVERA SM, HWANG JK, et al.Pharmacokinetics of mycophenolic acid after intravenous administration of mycophenolate mofetil to healthy cats[J].J Vet Intern Med, 2017, 31(6):1827-1832.DOI: 10.1111/jvim.14827. [4] ZHANG D, CHOW DS.Clinical pharmacokinetics of mycophenolic acid in hematopoietic stem cell transplantation recipients[J].Eur J Drug Metab Pharmacokinet, 2017, 42(2):183-189.DOI: 10.1007/s13318-016-0378-6. [5] RUSCHEL LR, SCHMITT VM, SILVA AB, et al.Study on the association of UGT1A9 gene c.98T > C polymorphism and mycophenolic acid plasma levels in renal transplant patients[J].Genet Mol Res, 2017, 16(2).DOI: 10.4238/gmr16029598. [6] 李朋梅, 张相林, 唐崑, 等.人血浆中霉酚酸浓度测定方法研究及药物监测应用[J].中国药学杂志, 2007, 42(19):1490-1493.DOI:10.3321/j.issn:1001- 2494.2007.19.015.LI PM, ZHANG XL, TANG K, et al.Determination of mycophenolic acid concentration in human plasma and its application to therapeutic drug monitoring in renal transplantation patient[J].Chin Pharm J, 2007, 42(19):1490-1493.DOI: 10.3321/j.issn:1001-2494.2007.19.015. [7] JEONG H, KAPLAN B.Therapeutic monitoring of mycophenolate mofetil[J].Clin J Am Soc Nephrol, 2007, 2(1):184-191. doi: 10.1097-01.tp.0000186380.61251.fc/ [8] MATHEW BS, FLEMING DH, ANNAPANDIAN VM, et al.A reliable limited sampling strategy for the estimation of mycophenolic acid area under the concentration time curve in adult renal transplant patients in the stable posttransplant period[J].Ther Drug Monit, 2010, 32(2):136-140.DOI: 10.1097/FTD.0b013e3181cd550f. [9] JIA Y, PENG B, LI L, et al.Estimation of mycophenolic acid area under the curve with limited-sampling strategy in Chinese renal transplant recipients receiving enteric-coated mycophenolate sodium[J].Ther Drug Monit, 2017, 39(1):29-36.DOI: 10.1097/FTD.0000000000000360. [10] LIU W, KULKARNI K, HU M.Gender-dependent differences in uridine 5'-diphospho-glucuronosyltransferase have implications in metabolism and clearance of xenobiotics[J].Expert Opin Drug Metab Toxicol, 2013, 9(12):1555-1569.DOI: 10.1517/17425255.2013.829040. [11] WU B, KULKARNI K, BASU S, et al.First-pass metabolism via UDP-glucuronosyltransferase:a barrier to oral bioavailability of phenolics[J].J Pharm Sci, 2011, 100(9):3655-3681.DOI: 10.1002/jps.22568. [12] 谢晓纯, 王洪阳, 李嘉丽, 等.性别对肾移植患者霉酚酸酯药代动力学影响的研究[J].中国临床药理学杂志, 2015, 31(6):456-458, 462.DOI: 10.13699/j.cnki.1001-6821.2015.06.017.XIE XC, WANG HY, LI JL, et al.Influence of gender on mycophenolate mofetil pharmacokinetics in Chinese renal transplant patients[J].Chin J Clin Pharmacol, 2015, 31(6):456-458, 462.DOI: 10.13699/j.cnki.1001-6821.2015.06.017. [13] 张丽娟, 陈璐, 朱宇轩, 等.有限采样法用于肾移植受者霉酚酸酯血药浓度监测的分析[J].中国生化药物杂志, 2016, 36(7):187-190.DOI:10.3969/j.issn.1005- 1678.2016.07.57.ZHANG LJ, CHEN L, ZHU YX, et al.Use of limited sampling strategy to analyse MMF blood concentration in renal transplant recipients[J].Chin J Biochem Pharmac, 2016, 36(7):187-190.DOI: 10.3969/j.issn.1005-1678.2016.07.57. [14] WOILLARD JB, REROLLE JP, PICARD N, et al.Risk of diarrhoea in a long-term cohort of renal transplant patients given mycophenolate mofetil:the significant role of the UGT1A8 2 variant allele[J].Br J Clin Pharmacol, 2010, 69(6):675-683.DOI: 10.1111/j.1365-2125.2010.03625.x. [15] ALVAREZ-ELÍAS AC, YOO EC, TODOROVA EK, et al.A retrospective study on mycophenolic acid drug interactions:effect of prednisone, sirolimus, and tacrolimus with MPA[J].Ther Drug Monit, 2017, 39(3):220-228.DOI: 10.1097/FTD.0000000000000403. -





 下载:
下载: