Experimental study of protective effect of hypothermic reperfusion on renal ischemia-reperfusion injury in rabbits
-
摘要:
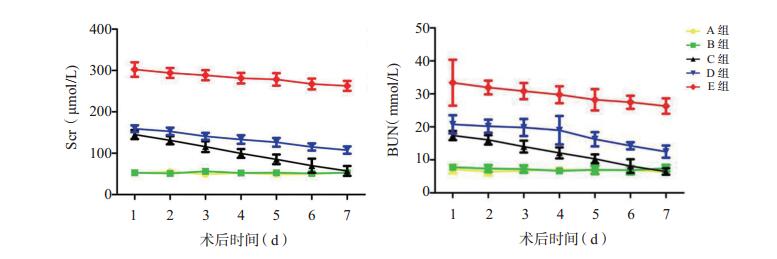
目的 探讨建立兔肾缺血低温环境、常温环境及高温环境再灌注损伤模型的新方法, 并评价低温环境复灌对兔肾缺血-再灌注损伤(IRI)的影响。 方法 将60只健康新西兰兔随机分为5组:对照组(A组)、假手术组(B组)、低温环境复灌组(C组)、常温环境复灌组(D组)、高温环境复灌组(E组), 每组12只。术后7 d内每日检测各组兔的血清肌酐(Scr)、血尿素氮(BUN)水平; 术后1 d检测各组兔肾组织内丙二醛(MDA)含量和超氧化物歧化酶(SOD)活性; 术后1 d采用苏木素-伊红(HE)染色观察肾组织病理学变化; 术后1 d采用dUTP缺口末端标记(TUNEL)染色评价细胞凋亡。 结果 术后1 d, 与A组和B组比较, C、D和E组兔的Scr和BUN水平均升高(均为P < 0.01);与C组比较, D组和E组兔的Scr和BUN水平升高更明显(均为P < 0.05)。术后7 d内, C、D和E组兔的Scr和BUN水平呈下降趋势。与D组和E组比较, C组兔的Scr和BUN水平较低(均为P < 0.05)。与A组和B组比较, C、D和E组的MDA含量均升高, SOD活性均降低(均为P < 0.01);与C组比较, D组和E组的MDA含量升高更明显, SOD活性更低(均为P < 0.01)。术后1 d肾组织病理学检查示A组和B组肾组织形态结构正常, D组和E组损伤表现明显, 与D、E组比较, C组损伤较轻。TUNEL染色结果显示, D组和E组肾小管上皮细胞阳性细胞明显增多, 管腔内也可见到阳性细胞, C组阳性细胞数量较D组和E组明显减少。 结论 冰泥覆盖肾脏、37 ℃生理盐水及40 ℃生理盐水连续滴加肾脏可建立不同温度环境复灌模型。低温环境复灌对肾IRI具有保护作用。 Abstract:Objective To explore a novel method for establishing rabbit models with renal ischemia-reperfusion injury (IRI) under hypothermic, normothermic and hyperthermic environments and evaluate the effect of the reperfusion under hypothermic environment on renal IRI in rabbits. Methods Sixty healthy New Zealand rabbits were randomly and evenly divided into five groups:control group (A group, n=12), sham operation group (B group, n=12), hypothermic reperfusion group (C group, n=12), normothermic reperfusion group (D group, n=12) and hyperthermic reperfusion group (E group, n=12).The serum creatinine (Scr) and blood urea nitrogen (BUN) levels were measured daily within postoperative 7 d.The content of malondialdehyde (MDA) and the activity of superoxide dismutase (SOD) in the renal tissues were measured at postoperative 1 d.Hematoxylin-eosin (HE) staining was performed to observe the histopathological changes at postoperative 1 d.Terminal deoxynucleotidyl transferase (TdT)-dUTP nick-end labeling (TUNEL staining) was adopted to detect the cellular apoptosis at postoperative 1 d. Results At 1 d after operation, the Scr and BUN levels in C, D and E groups were significantly higher than those in A and B groups (all P < 0.01).Compared with C group, the Scr and BUN levels were more notably increased in D and E groups (both P < 0.05).Within 7 d after surgery, the Scr and BUN levels tended to decline in C, D and E groups.Compared with D and E groups, the Scr and BUN levels were significantly lower in C group (both P < 0.05).Compared with A and B groups, the MDA contents were considerably increased, whereas the SOD activities were significantly decreased in C, D and E groups (all P < 0.01).Compared with C group, the MDA content was more significantly enhanced, whereas the SOD activity was remarkably decreased in D and E groups (both P < 0.01).At postoperative 1 d, the pathological examination of the renal tissues demonstrated that the renal morphology was normal in A and B groups, whereas significant renal damage was observed in D and E groups.Compared with D and E groups, the severity of renal injury was less in C group.TUNEL staining revealed that the quantity of positive renal tubular epithelial cells was significantly increased in D and E groups, and positive cells were also seen in the lumen.The quantity of positive cells in C group was significantly lower than those in D and E groups. Conclusions The reperfusion rabbit models under different temperature environments can be established by covering the kidney by ice soil and persistent administration of 37 ℃ and 40 ℃ saline.Hypothermic reperfusion exerts a protective effect on renal IRI. -
表 1 术后1 d各组兔肾组织MDA和SOD水平的比较
Table 1. Comparison of MDA and SOD levels in renal tissues of rabbits among each group at 1 d after operation(x±s)
组别 n MDA(nmol/mg prot) SOD(U/mg prot) A组 6 1.30±0.06 618±35 B组 6 1.22±0.09 623±29 C组 6 2.44±0.18a, b 495±34a, b D组 6 3.45±0.34a, b, c 409±32a, b, c E组 6 6.96±0.36a, b, c 203±47a, b, c 与A组比较, aP < 0.01;与B组比较, bP < 0.01;与C组比较, cP < 0.01 -
[1] ZUK A, BONVENTRE JV.Acute Kidney Injury[J].Annu Rev Med, 2016, 67:293-307.DOI: 10.1146/annurev-med-050214-013407. [2] PERICO N, CATTANEO D, SAYEGH MH, et al.Delayed graft function in kidney transplantation[J].Lancet, 2004, 364(9447):1814-1827. doi: 10.1016/S0140-6736(04)17406-0 [3] YANG Y, SONG M, LIU Y, et al.Renoprotective approaches and strategies in acute kidney injury[J].Pharmacol Ther, 2016, 163:58-73.DOI: 10.1016/j.pharmthera.2016.03.015. [4] NIEMANN CU, FEINER J, SWAIN S, et al.Therapeutic hypothermia in deceased organ donors and kidney-graft function[J].N Engl J Med, 2015, 373(5):405-414.DOI: 10.1056/NEJMoa1501969. [5] CHENOUNE M, LIDOUREN F, ADAM C, et al.Ultrafast and whole-body cooling with total liquid ventilation induces favorable neurological and cardiac outcomes after cardiac arrest in rabbits[J].Circulation, 2011, 124(8):901-911, 1-7.DOI: 10.1161/CIRCULATIONAHA.111.039388. [6] 林亚平, 刘琴, 陈楚淘, 等.针刺联合亚低温对脑缺血再灌注损伤大鼠脑组织MAPK/ERK通路及凋亡相关因子的影响[J].中南大学学报(医学版), 2017, 42(4):380-388.DOI: 10.11817/j.issn.1672-7347.2017.04.003.LIN YP, LIU Q, CHEN CT, et al.Effect of acupuncture combined with hypothermia on MAPK/ERK pathway and apoptosis related factors in rats with cerebral ischemia reperfusion injury[J].J Centr South Univ(Med Sci), 2017, 42(4):380-388.DOI: 10.11817/j.issn.1672-7347.2017.04.003. [7] FENG JZ, WANG WY, ZENG J, et al.Optimization of brain metabolism using metabolic-targeted therapeutic hypothermia can reduce mortality from traumatic brain injury[J].J Trauma Acute Care Surg, 2017, 83(2):296-304.DOI: 10.1097/TA.0000000000001522. [8] TISSIER R, COHEN MV, DOWNEY JM.Does mild hypothermia protect against reperfusion injury? the debate continues[J].Basic Res Cardiol, 2011, 106(5):691-695.DOI: 10.1007/s00395-011-0194-8. [9] GOLDFARB DA.Re:therapeutic hypothermia in deceased organ donors and kidney-graft function[J].J Urol, 2016, 195(5):1549.DOI: 10.1016/j.juro.2016.02.010. [10] 余晓东, 廖波, 邓显忠, 等.一种新型实用的大鼠肾缺血再灌注损伤模型的建立[J].重庆医学, 2011, 40(13):1283-1284, 封3-封4.DOI: 10.3969/j.issn.1671-8348.2011.13.014.YU XD, LIAO B, DENG XZ, et al.Model construction of renal ischemia-reperfusion injury in rats[J].Chongqing Med, 2011, 40(13):1283-1284, cover3-4.DOI: 10.3969/j.issn.1671-8348.2011.13.014. [11] SALVADORI M, ROSSO G, BERTONI E.Update on ischemia-reperfusion injury in kidney transplantation:pathogenesis and treatment[J].World J Transplant, 2015, 5(2):52-67.DOI: 10.5500/wjt.v5.i2.52. [12] DELBRIDGE MS, SHRESTHA BM, RAFTERY AT, et al.The effect of body temperature in a rat model of renal ischemia-reperfusion injury[J].Transplant Proc, 2007, 39(10):2983-2985. doi: 10.1016/j.transproceed.2007.04.028 [13] 蔺艳, 何涛, 毛晓燕, 等.还原型谷胱甘肽对肾急性缺血再灌注性损伤的作用[J].实用医学杂志, 2016, 32(8):1233-1236.DOI:10.3969/j.issn.1006-5725.2016. 08.010.LIN Y, HE T, MAO XY, et al.Protective effect of reduced glutathione on kidney against acute ischemia-reperfusion injury[J].J Pract Med, 2016, 32(8):1233-1236.DOI: 10.3969/j.issn.1006-5725.2016.08.010. [14] AKHTAR MZ, SUTHERLAND AI, HUANG H, et al.The role of hypoxia-inducible factors in organ donation and transplantation:the current perspective and future opportunities[J].Am J Transplant, 2014, 14(7):1481-1487.DOI: 10.1111/ajt.12737. [15] AYODELE M, KOCH S.Ischemic preconditioning in the intensive care unit[J].Curr Treat Options Neurol, 2017, 19(6):24.DOI: 10.1007/s11940-017-0457-2. [16] 艾娜, 谢席胜, 樊均明, 等.大鼠肾缺血再灌注损伤模型改良手术方式结果与评价[J].中国中西医结合肾病杂志, 2013, 14(2):104-106, 后插2.DOI: 10.3969/j.issn.1009-587X.2013.02.004.AI N, XIE XS, FAN JM, et al.The Improved model of the operation mode of renal ischemia reperfusion injury in rats and evaluation[J].Chin J Integr Trad West Nephrol, 2013, 14(2):104-106, back insertion 2.DOI: 10.3969/j.issn.1009-587X.2013.02.004. [17] 韦星, 蔡明, 石炳毅, 等.新型肾脏冷缺血再灌注损伤动物模型的稳定性研究[J/CD].中华临床医师杂志(电子版), 2012, 6(5): 1207-1210.DOI: 10.3877/cma.j.issn.1674-0785.2012.05.030.WEI X, CAI M, SHI BY, et al.Stability of a new model of kidney cold ischemia reperfusion injury[J/CD].Chin J Clin (Electr Edit), 2012, 6(5): 1207-1210.DOI: 10.3877/cma.j.issn.1674-0785.2012.05.030. [18] PALLER MS, HOIDAL JR, FERRIS TF.Oxygen free radicals in ischemic acute renal failure in the rat[J].J Clin Invest, 1984, 74(4):1156-1164. doi: 10.1172/JCI111524 -





 下载:
下载: