-
摘要:
目的 探讨FAM234A是否为一种新的非Gal异种移植抗原。 方法 用α-1, 3-半乳糖基转移酶基因敲除(GTKO)小型猪的原代猪主动脉内皮细胞(PAEC)免疫食蟹猴。将免疫的猴血清与PAEC孵育, 其中经免疫产生的猴抗猪细胞抗体能够识别并结合PAEC表面的未知抗原。采用流式细胞术测定PAEC表面结合的猴抗猪细胞抗体的平均荧光强度(MFI), 用以表示PAEC表面抗原的含量。利用慢病毒介导的短发夹核糖核酸(shRNA)敲减PAEC中的FAM234A基因, 检测该细胞结合的猴抗猪细胞抗体含量是否减少, 以确定该基因是否为潜在的移植抗原。 结果 PAEC中的非Gal抗原能够在猴体内引发大量的猴抗猪细胞抗体。在GTKO小型猪的PAEC中用shRNA敲减FAM234A基因后, PAEC结合的猴IgG抗体减少。 结论 FAM234A是一种新的非Gal异种移植抗原。 -
关键词:
- 异种移植 /
- 非Gal抗原 /
- 主动脉内皮细胞(PAEC) /
- FAM234A /
- 食蟹猴 /
- α-1, 3-半乳糖基转移酶基因敲除(GTKO) /
- 流式细胞术 /
- 短发夹核糖核酸(shRNA) /
- 基因敲减 /
- 巴马小型猪 /
- 五指山小型猪 /
- 慢病毒
Abstract:Objective To investigate whether FAM234A is a novel non-Gal antigen in xenotransplantation. Methods Cynomolgus monkeys were immunized with primary porcine aorta endothelial cells (PAEC) from α-1, 3-galactosyltransferase gene-knockout (GTKO) minipigs.The serum samples of immunized monkeys were incubated with PAEC.After immunization, the monkey anti-pig cell antibodies were able to recognize and bind with the unknown antigens on the PAEC surface.Flow cytometry was adopted to determine the mean fluorescence intensity (MFI) of monkey anti-pig cell antibodies binding with the PAEC surface to characterize the content of PAEC surface antigen.Lentivirus-mediated short hairpin ribonucleic acid (shRNA) was utilized to knock down the FAM234A gene in PAEC to detect whether the content of the cell-bound monkey anti-pig cell antibody was reduced and determine if this gene was a potential transplant antigen. Results The non-Gal antigen in PAEC could trigger a large quantity of monkey anti-pig cell antibodies in monkeys.After knock down of the FAM234A gene with shRNA in the PAEC of the GTKO miniature pigs, the monkey IgG antibody binding with PAEC was declined. Conclusions FAM234A is a novel non-Gal antigen in xenotransplantation. -
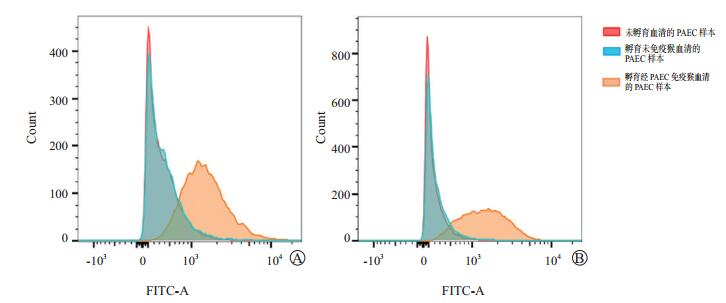
图 3 敲减FAM234A使PAEC与免疫猴血清中的IgG结合减少
A图为五指山小型猪; B图为巴马小型猪; 左侧为抗猪细胞抗体的流式细胞图, 中间为流式细胞参数图, 右侧为不同样本结合抗体的MFI及FAM234A mRNA水平的比较。红色为未孵育血清的PAEC样本, 蓝色为孵育经PAEC免疫猴血清的对照PAEC样本(plko), 黄色为孵育经PAEC免疫猴血清的敲减FAM234A的PAEC样本(plko-FAM234A)
Figure 3. Knock down of FAM234A in PAECs results in a reduction in binding of serum IgG fromimmunized monkey
-
[1] COOPER DKC, GASTON R, ECKHOFF D, et al.Xenotransplantation-the current status and prospects[J].Br Med Bull, 2018, 125(1):5-14.DOI: 10.1093/bmb/ldx043. [2] MATSUDA Y, SARWAL MM.Unraveling the role of allo-antibodies and transplant injury[J].Front Immunol, 2016, 7:432. https://www.ncbi.nlm.nih.gov/pubmed/27818660 [3] COOPER DK, EZZELARAB MB, HARA H, et al.The pathobiology of pig-to-primate xenotransplantation:a historical review[J].Xenotransplantation, 2016, 23(2):83-105.DOI: 10.1111/xen.12219. [4] COOPER DKC, EKSER B, TECTOR AJ.Immunobiological barriers to xenotransplantation[J].Int J Surg, 2015, 23(Pt B):211-216.DOI: 10.1016/j.ijsu.2015.06.068. [5] BYRNE GW, MCGREGOR CGA, BREIMER ME.Recent investigations into pig antigen and anti-pig antibody expression[J].Int J Surg, 2015, 23(Pt B):223-228.DOI: 10.1016/j.ijsu.2015.07.724. [6] DAI Y, VAUGHT TD, BOONE J, et al.Targeted disruption of the alpha1, 3-galactosyltransferase gene in cloned pigs[J].Nat Biotechnol, 2002, 20(3):251-255. doi: 10.1038/nbt0302-251 [7] PHELPS CJ, KOIKE C, VAUGHT TD, et al.Production of alpha 1, 3-galactosyltransferase-deficient pigs[J].Science, 2003, 299(5605):411-414. doi: 10.1126/science.1078942 [8] LUTZ AJ, LI P, ESTRADA JL, et al.Double knockout pigs deficient in N-glycolylneuraminic acid and galactose α-1, 3-galactose reduce the humoral barrier to xenotransplantation[J].Xenotransplantation, 2013, 20(1):27-35.DOI: 10.1111/xen.12019. [9] COOPER DK, EKSER B, RAMSOONDAR J, et al.The role of genetically engineered pigs in xenotransplantation research[J].J Pathol, 2016, 238(2):288-299.DOI: 10.1002/path.4635. [10] CHEN G, QIAN H, STARZL T, et al.Acute rejection is associated with antibodies to non-Gal antigens in baboons using Gal-knockout pig kidneys[J].Nat Med, 2005, 11(12):1295-1298. doi: 10.1038/nm1330 [11] MARTENS GR, REYES LM, BUTLER JR, et al.Humoral reactivity of renal transplant-waitlisted patients to cells from GGTA1/CMAH/B4GalNT2, and SLA class Ⅰ knockout pigs[J].Transplantation, 2017, 101(4):e86-e92.DOI: 10.1097/TP.0000000000001646. [12] LI JH, ZHAO B, ZHU XH, et al.Blockade of extracellular HMGB1 suppresses xenoreactive B cell responses and delays acute vascular xenogeneic rejection[J].Am J Transplant, 2015, 15(8):2062-2074.DOI: 10.1111/ajt.13275. [13] EKSER B, LI P, COOPER DKC.Xenotransplantation:past, present, and future[J].Curr Opin Organ Transplant, 2017, 22(6):513-521.DOI: 10.1097/MOT.0000000000000463. [14] WOLLSCHEID B, BAUSCH-FLUCK D, HENDERSON C, et al.Mass-spectrometric identification and relative quantification of N-linked cell surface glycoproteins[J].Nat Biotechnol, 2009, 27(4):378-386.DOI: 10.1038/nbt.1532. [15] CHEN Z, TANG H, QAYYUM R, et al.Genome-wide association analysis of red blood cell traits in African Americans:the COGENT Network[J].Hum Mol Genet, 2013, 22(12):2529-2538.DOI: 10.1093/hmg/ddt087. [16] ZHANG J, XIE C, LU Y, et al.Potential antigens involved in delayed xenograft rejection in a Ggta1/Cmah Dko pig-to-monkey model[J].Sci Rep, 2017, 7(1):10024.DOI: 10.1038/s41598-017-10805-0. [17] ESTRADA JL, MARTENS G, LI P, et al.Evaluation of human and non-human primate antibody binding to pig cells lacking GGTA1/CMAH/β4GalNT2 genes[J].Xenotransplantation, 2015, 22(3):194-202.DOI: 10.1111/xen.12161. -





 下载:
下载: