Comparison of clinical efficacy between ABO-incompatible and ABO-compatible liver transplantation in children: a Meta-analysis
-
摘要:
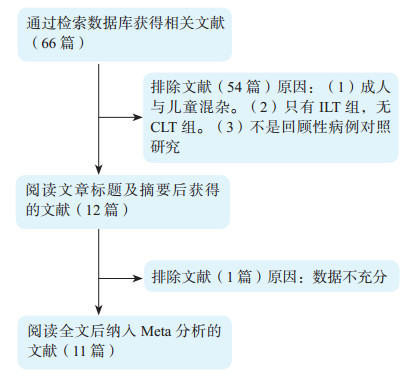
目的 应用Meta分析对儿童ABO血型不合肝移植(ILT)与儿童ABO血型相合肝移植(CLT)的疗效进行比较。 方法 检索PubMed、Embase、Cochrane library、中国知网、万方、维普等国内外数据库,检索时间截至2017年5月。根据制定的纳入标准和排除标准筛选符合要求的文献,提取数据。采用Review Manager 5.3软件,应用随机效应模型或固定效应模型进行Meta分析。 结果 共纳入11篇英文文献,均为回顾性队列研究。Meta分析结果表明ILT组受者术后1年存活率明显低于CLT组[比值比(OR)=0.64,95%可信区间(CI)0.49~0.83,P=0.0007)];ILT组术后排斥反应发生率明显高于CLT组(OR=1.96,95% CI 1.03~3.72,P=0.04);两组的术后3、10年受者存活率,术后1、3、10年移植物存活率,术后胆道并发症发生率差异无统计学意义(均为P > 0.05)。 结论 与CLT比较,ILT受者1年存活率较低而排斥反应发生率较高,但两组受者和移植物长期存活率及胆道并发症发生率相似。表明ILT是在紧急情况下挽救患者生命或长时间无法等到血型相合的肝源时较为理想的选择。 Abstract:Objective To compare the clinical efficacy between pediatric ABO-incompatible liver transplantation (ILT) and ABO-compatible liver transplantation (CLT) by Meta-analysis. Methods Relevant studies published until May 2017 were electronically retrieved from PubMed, Embase, Cochrane library, China national knowledge internet (CNKI), Wanfang and VIP databases. According to the inclusion and exclusion criteria, the publications eligible were screened and clinical data were extracted. Meta-analysis was performed using the random or fixed effect model analyses with Review Manager 5.3 statistical software. Results Eleven retrospective cohort studies in English were selected. Meta-analysis demonstrated that the postoperative 1-year survival rate of the recipients in the ILT group was significantly lower than that in the CLT group [odds ratio (OR)=0.64, 95% confidence interval (CI) 0.49-0.83, P=0.0007)]. In the ILT group, the incidence of postoperative rejection reactions was considerably higher compared with that in the CLT group (OR=1.96, 95% CI 1.03-3.72, P=0.04). No statistical significance was observed in the postoperative 3-and 10-year survival rate of the recipients, 1-, 3-and 10-year survival rate of the graft and the incidence of postoperative biliary tract complications between two groups (all P > 0.05). Conclusions Compared with their CLT counterparts, the 1-year survival rate of the ILT recipients is lower, whereas the incidence of rejection reactions is higher. However, the long-term survival rate of both the recipient and graft and the incidence of biliary tract complications do not significantly differ between CLT and ILT. ILT is a relatively ideal option for emergent patients or those lacking of liver graft with compatible blood group for a long period of time. -
表 1 纳入11篇文献的基本情况
Table 1. Basic condition of 11 studies included
究作者 年份 国家 NOS评分 ILT组 CLT组 总例数 1、3、10年受者存活例数 1、3、10年移植物存活例数 胆道并发症发生例数 排斥反应发生例数 总例数 1、3、10年受者存活例数 1、3、10年移植物存活例数 胆道并发症发生例数 排斥反应发生例数 Schukfeh N, et al[10] 2015 德国 7 6 5/-/- 5/-/- 0 1 40 40/-/- 33/-/- - - Kasahara M, et al[11] 2013 日本 8 294 239/-/220 - - - 1 930 1 724/-/1 620 - - - Gelas T, et al[12] 2011 英国 7 5 5/-/- 5/-/- 0 3 24 15/-/- 15/-/- 4 4 Heffron TG, et al[13] 2010 美国 8 12 11/9/- 11/9/- - 6 21 20/20/- 20/17/- - 7 Stewart ZA, et al[14] 2009 美国 8 246 - 164/151/132 - - 738 - 497/461/418 - - Ueda M, et al[15] 2006 日本 8 74 58/-/47 57/-/46 - - 494 423/-/390 421/-/337 - - Heffron T, et al[16] 2006 美国 7 16 16/-/- 15/-/- 2 6 122 113/-/- 102/-/- 13 47 Varela-Fascinetto G, et al[17] 1999 美国 8 28 20/20/20 -/-/19 - - 72 48/46/44 -/-/38 - - Cacciarelli TV, et al[18] 1995 美国 8 14 11/11/- 10/10/- 1 11 130 109/102/- 109/102/- 7 73 Yandza T, et al[19] 1994 美国 7 7 5/-/- 4/-/- 0 1 93 57/-/- 56/-/- 16 - Tokunaga Y, et al[4] 1993 美国 7 3 3/-/- 3/-/- 0 1 31 27/-/- 27/-/- 3 3 -表示文献中未具体提及或数据难以提取 -
[1] Sanchez-Urdazpal L, Batts KP, Gores GJ, et al. Increased bile duct complications in liver transplantation across the ABO barrier[J]. Ann Surg, 1993, 218(2): 152-158. doi: 10.1097/00000658-199308000-00006 [2] Gugenheim J, Samuel D, Reynes M, et al. Liver transplantation across ABO blood group barriers[J]. Lancet, 1990, 336(8714): 519-523. doi: 10.1016/0140-6736(90)92082-S [3] Fischel RJ, Ascher NL, Payne WD, et al. Pediatric liver transplantation across ABO blood group barriers[J]. Transplant Proc, 1989, 21(1 Pt 2): 2221-2222. [4] Tokunaga Y, Tanaka K, Fujita S, et al. Living related liver transplantation across ABO blood groups with FK506 and OKT3[J]. Transpl Int, 1993, 6(6): 313-318. doi: 10.1111/j.1432-2277.1993.tb00673.x [5] Okada N, Sanada Y, Hirata Y, et al. The impact of rituximab in ABO-incompatible pediatric living donor liver transplantation: the experience of a single center[J]. Pediatr Transplant, 2015, 19(3): 279-286. DOI: 10.1111/petr.12445. [6] Sanada Y, Mizuta K, Urahashi T, et al. Role of apheresis and dialysis in pediatric living donor liver transplantation: a single center retrospective study[J]. Ther Apher Dial, 2012, 16(4): 368-375. DOI: 10.1111/j.1744-9987.2012.01079.x. [7] Kozaki K, Egawa H, Kasahara M, et al. Therapeutic strategy and the role of apheresis therapy for ABO incompatible living donor liver transplantation[J]. Ther Apher Dial, 2005, 9(4): 285-291. DOI: 10.1111/j.1744-9987.2005.00304.x. [8] Hashimoto T, Kondo S, Suzuki T, et al. Strategy for ABO-incompatible living-related liver transplantation[J]. Transplant Proc, 2000, 32(7): 2104-2106. doi: 10.1016/S0041-1345(00)01589-X [9] Rana A, Kueht ML, Nicholas SK, et al. Pediatric liver transplantation across the ABO blood group barrier: is it an obstacle in the modern era?[J]. J Am Coll Surg, 2016, 222(4): 681-689. DOI: 10.1016/j.jamcollsurg.2015.12.041. [10] Schukfeh N, Lenz V, Metzelder ML, et al. First case studies of successful ABO-incompatible living-related liver transplantation in infants in Germany[J]. Eur J Pediatr Surg, 2015, 25(1): 77-81. DOI: 10.1055/s-0034-1387936. [11] Kasahara M, Umeshita K, Inomata Y, et al. Long-term outcomes of pediatric living donor liver transplantation in Japan: an analysis of more than 2200 cases listed in the Registry of the Japanese Liver Transplantation Society[J]. Am J Transplant, 2013, 13(7): 1830-1839. DOI: 10.1111/ajt.12276. [12] Gelas T, McKiernan PJ, Kelly DA, et al. ABO-incompatible pediatric liver transplantation in very small recipients: Birmingham' s experience[J]. Pediatr Transplant, 2011, 15(7): 706-711. DOI: 10.1111/j.1399-3046.2011.01541.x. [13] Heffron TG, Pillen T, Smallwood G, et al. Pediatric liver transplantation for acute liver failure at a single center: a 10-yr experience[J]. Pediatr Transplant, 2010, 14(2): 228-232. DOI: 10.1111/j.1399-3046.2009.01202.x. [14] Stewart ZA, Locke JE, Montgomery RA, et al. ABO-incompatible deceased donor liver transplantation in the United States: a national registry analysis[J]. Liver Transpl, 2009, 15(8):883-893. DOI: 10.1002/lt.21723. [15] Ueda M, Oike F, Ogura Y, et al. Long-term outcomes of 600 living donor liver transplants for pediatric patients at a single center[J]. Liver Transpl, 2006, 12(9): 1326-1336. DOI: 10.1002/lt.20826. [16] Heffron T, Welch D, Pillen T, et al. Successful ABO-incompatible pediatric liver transplantation utilizing standard immunosuppression with selective postoperative plasmapheresis[J]. Liver Transpl, 2006, 12(6): 972-978. DOI: 10.1002/lt.20760. [17] Varela-Fascinetto G, Treacy SJ, Lillehei CW, et al. Long-term results in pediatric ABO-incompatible liver transplantation[J]. Transplant Proc, 1999, 31(1/2): 467-468. doi: 10.1016-S0041-1345(98)01711-4/ [18] Cacciarelli TV, So SK, Lim J, et al. A reassessment of ABO incompatibility in pediatric liver transplantation[J]. Transplantation, 1995, 60(7): 757-760. doi: 10.1097/00007890-199510150-00024 [19] Yandza T, Gauthier F, Valayer J. Lessons from the first 100 liver transplantations in children at Bicêtre hospital[J]. J Pediatr Surg, 1994, 29(7): 905-911. doi: 10.1016/0022-3468(94)90013-2 [20] Haga H, Egawa H, Fujimoto Y, et al. Acute humoral rejection and C4d immunostaining in ABO blood type-incompatible liver transplantation[J]. Liver Transpl, 2006, 12(3): 457-464. DOI: 10.1002/lt.20652. [21] Egawa H, Teramukai S, Haga H, et al. Impact of rituximab desensitization on blood-type-incompatible adult living donor liver transplantation: a Japanese multicenter study[J]. Am J Transplant, 2014, 14(1): 102-114. DOI: 10.1111/ajt.12520. [22] Tessier ME, Harpavat S, Shepherd RW, et al. Beyond the pediatric end-stage liver disease system: solutions for infants with biliary atresia requiring liver transplant[J]. World J Gastroenterol, 2014, 20(32): 11062-11068. DOI: 10.3748/wjg.v20.i32.11062. [23] Fong SW, Qaqundah BY, Taylor WF. Developmental patterns of ABO isoagglutinins in normal children correlated with the effects of age, sex, and maternal isoagglutinins[J]. Transfusion, 1974, 14(6): 551-559. doi: 10.1111/trf.1974.14.issue-6 [24] Ferriani VP, Barbosa JE, de Carvalho IF. Serum haemolytic classical and alternative pathways of complement in infancy: age-related changes[J]. Acta Paediatr Scand, 1990, 79(3): 322-327. doi: 10.1111/apa.1990.79.issue-3 [25] Egawa H, Oike F, Buhler L, et al. Impact of recipient age on outcome of ABO-incompatible living-donor liver transplantation[J]. Transplantation, 2004, 77(3): 403-411. DOI: 10.1097/01.TP.0000110295.88926.5C. [26] Yandza T, Lambert T, Alvarez F, et al. Outcome of ABO-incompatible liver transplantation in children with no specific alloantibodies at the time of transplantation[J].Transplantation, 1994, 58(1): 46-50. doi: 10.1097/00007890-199407000-00009 [27] Hussein MH, Hashimoto T, AbdEl-Hamid Daoud G, et al. Pediatric patients receiving ABO-incompatible living related liver transplantation exhibit higher serum transforming growth factor-β1, interferon-γ and interleakin-2 levels[J]. Pediatr Surg Int, 2011, 27(3): 263-268. DOI: 10.1007/s00383-010-2784-1. -





 下载:
下载: