Induction of Schwann cell-like differentiation in bone mesenchymal stem cell of adult rat in vitro
-
摘要:
目的 探讨体外定向诱导成年大鼠骨髓间充质干细胞(BMSC)向许旺细胞样细胞转化的有效方法。 方法 分别进行成年大鼠许旺细胞原代培养和BMSC分离培养。根据诱导方法不同, 分为化学诱导组和共培养诱导组。采用显微镜观察许旺细胞和BMSC的生长情况, 分别采用免疫荧光化学染色法[检测神经胶质纤维酸性蛋白(GFAP)抗体和S-100抗体等许旺细胞标志蛋白]和流式细胞术鉴定两种细胞; 显微镜下动态观察两组细胞的形态和生长情况; 共培养诱导组共培养3 d后, 化学诱导组诱导作用4 h、1 d后分别采用免疫荧光化学染色的方法评估两组细胞的诱导分化情况。 结果 化学诱导组诱导后的细胞发生了明显的许旺细胞样细胞形态改变, 预诱导4 h后即出现GFAP抗体表达阳性, 维持诱导1 d后, GFAP抗体阳性表达率为(80.9±3.5)%, S-100抗体阳性表达率为(59.0±1.1)%。诱导2 d后分化细胞开始出现脱落死亡, 3 d后大部分分化细胞脱落死亡。共培养诱导组共培养3 d后, 被诱导的BMSC未出现类似化学诱导组明显的细胞形态改变, GFAP抗体阳性表达率为(89.8±2.4)%, S-100抗体阳性表达率为(80.9±1.7)%。共培养诱导组S-100、GFAP抗体阳性表达率均高于化学诱导组, 差异均有统计学意义(均为P < 0.01)。 结论 共培养诱导法不仅对BMSC向许旺细胞样细胞定向分化具有明确的效果, 而且对BMSC的存活和增殖均有促进作用, 因此该法相对于化学诱导法更加安全和有效。 Abstract:Objective To explore the effective method of induction of Schwann cell-like differentiation in bone mesenchymal stem cell (BMSC) of adult rat in vitro. Methods Primary culture of Schwann cell and isolated culture of BMSC were separately conducted. According to different induction methods, the cells were divided into chemical induction group and co-culture induction group. The growth of Schwann cell and BMSC was observed under light microscope. These two kinds of cells were identified by immunofluorescence staining [detecting Schwann cell marker proteins: glial fibrillary acidic protein (GFAP) antibody and S-100 antibody] and flow cytometry. The shape and growth of cells in two groups were dynamically observed by light microscope. The induced differentiation was evaluated with immunofluorescence staining at 3rd day after co-culture induction in the co-culture induction group and at 4 h and 1st day after chemical induction in the chemical induction group. Results In the chemical induction group, the BMSC appeared typical Schwann cell-like morphology. The positive expression of GFAP antibody appeared at 4 h after preliminary induction. Meanwhile, the positive expression rate of GFAP and S-100 antibody was (80.9±3.5)% and (59.0±1.1)% at 1st day after induction. The induced BMSC began to die at 2nd day after chemical induction and most of the induced BMSC had died at 3rd day after chemical induction. At 3rd day after co-culture induction, few induced BMSC showed obvious morphological changes like those in chemical induction group. The positive expression rate of GFAP and S-100 antibody was (89.8±2.4)% and (80.9±1.7)%. The positive expression rate of GFAP and S-100 antibody in the co-culture induction group was higher than those in the chemical induction group and the difference had statistical significance (all in P < 0.01). Conclusions The co-culture induction not only has obvious effect on Schwann cell-like differentiation in BMSC, but also promotes the survival and proliferation of BMSC. Thus, co-culture induction is more safe and effective than chemical induction. -
Key words:
- Bone mesenchymal stem cell /
- Schwann cell /
- Co-culture /
- Induction /
- Differentiation
-
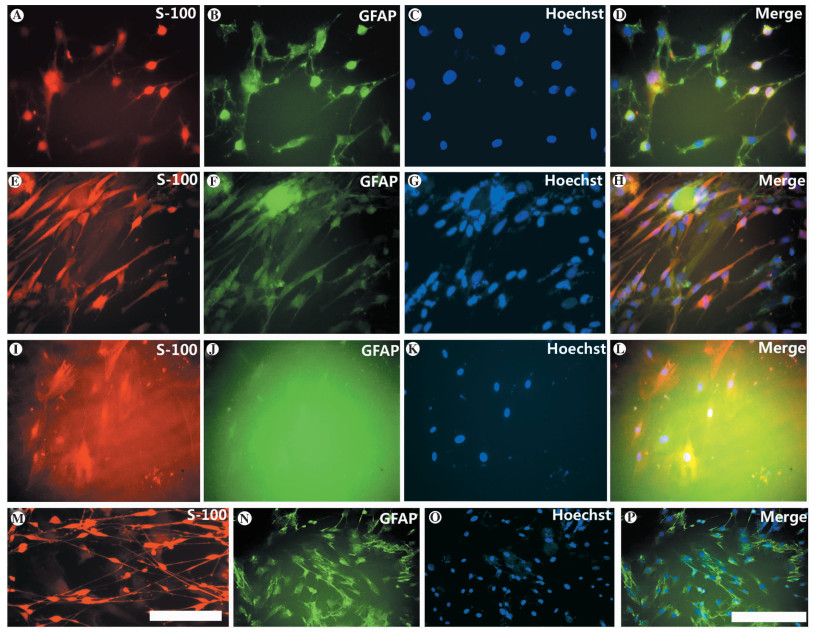
图 1 化学诱导法和共培养诱导法诱导成年大鼠骨髓间充质干细胞向许旺细胞样细胞分化的荧光鉴定图
注:A~D图为化学诱导组维持诱导液诱导1 d后,细胞中S-100、GFAP抗体的表达情况,其中A图为S-100抗体,B图为GFAP抗体,C图为Hoechst,D图为前三者重合图像(×200);E~H图为共培养诱导组共培养3 d后,细胞中S-100、GFAP抗体的表达情况,其中E图为S-100抗体,F图为GFAP抗体,G图为Hoechst,H图为前三者重合图像(×200);I~L图为对照组未经诱导的BMSC中S-100、GFAP抗体的表达情况,其中I图为S-100抗体,J图为GFAP抗体,K图为Hoechst,L图为前三者重合图像(×200);M图为原代培养的许旺细胞(S-100抗体,×200);N~P图为化学诱导组在预诱导4 h后, 被诱导的BMSC细胞形态未出现明显变化,但已经表达GFAP抗体,其中N图为GFAP抗体,O图为Hoechst,P图为前两者重合图像(×100)
Figure 1. Fluorescent identification figures of Schwann cell-like differentiation in BMSC of adult rat in vitro by co-culture with Schwann cells and chemical induction
-
[1] Erakat MS, Chuang SK, Shanti RM, et al. Interval between injury and lingual nerve repair as a prognostic factor for success using type Ⅰ collagen conduit[J]. J Oral Maxillofac Surg, 2013, 71(5):833-838. doi: 10.1016/j.joms.2011.11.026 [2] Liu XY, Chen J, Zhou Q, et al. In vitro tissue engineering of lamellar cornea using human amniotic epithelial cells and rabbit cornea stroma[J]. Int J Ophthalmol, 2013, 6(4):425-429. http://en.cnki.com.cn/Article_en/CJFDTOTAL-GYZZ201304003.htm [3] Mungadi IA. Bioengineering tissue for organ repair, regeneration, and renewal[J]. J Surg Tech Case Rep, 2012, 4(2):77-78. doi: 10.4103/2006-8808.110247 [4] Tang H, Wu B, Qin X, et al. Tissue engineering rib with the incorporation of biodegradable polymer cage and BMSCs/decalcified bone:an experimental study in a canine model[J]. J Cardiothorac Surg, 2013, 8:133. doi: 10.1186/1749-8090-8-133 [5] Azanchi R, Bernal G, Gupta R, et al. Combined demyelination plus Schwann cell transplantation therapy increases spread of cells and axonal regeneration following contusion injury[J]. J Neurotrauma, 2004, 21(6):775-788. doi: 10.1089/0897715041269696 [6] Novikov LN, Novikova LN, Mosahebi A, et al. A novel biodegradable implant for neuronal rescue and regeneration after spinal cord injury[J]. Biomaterials, 2002, 23(16):3369-3376. doi: 10.1016/S0142-9612(02)00037-6 [7] 万虹, 李德志, 杨飞, 等.许旺细胞与PLGA共同移植于大鼠全横断脊髓损伤的实验研究[J].中华外科杂志, 2007, 45(12):843-846. http://d.wanfangdata.com.cn/Periodical/zhwk200712016Wan H, Li DZ, Yang F, et al. Research about Schwann cells and PLGA implanted to rat transected spinal cord[J]. Chin J Surg, 2007, 45(12):843-846. http://d.wanfangdata.com.cn/Periodical/zhwk200712016 [8] Elias PZ, Spector M. Implantation of a collagen scaffold seeded with adult rat hippocampal progenitors in a rat model of penetrating brain injury[J]. J Neurosci Methods, 2012, 209(1):199-211. doi: 10.1016/j.jneumeth.2012.06.003 [9] Yu H, Cao B, Feng M, et al. Combinated transplantation of neural stem cells and collagen type I promote functional recovery after cerebral ischemia in rats[J]. Anat Rec, 2010, 293(5):911-917. doi: 10.1002/ar.20941 [10] Huang B, Tabata Y, Gao JQ. Mesenchymal stem cells as therapeutic agents and potential targeted gene delivery vehicle for brain diseases[J]. J Control Release, 2012, 162(2):464-473. doi: 10.1016/j.jconrel.2012.07.034 [11] 孙翀, 尹东亮, 朱焕斌, 等.改良全骨髓贴壁法分离与纯化大鼠骨髓间充质干细胞[J/CD].中华肝脏外科手术学电子杂志, 2014, 3(6):384-388. http://d.wanfangdata.com.cn/Periodical/zhgzwkssxdzzz201406013Sun C, Yin DL, Zhu HB, et al. Modified whole bone marrow adherent method for the isolation and purification of rats bone marrow mesenchymal stem cells[J/CD]. Chin J Hepat Surg:Electr Edit, 2014, 3(6):384-388. http://d.wanfangdata.com.cn/Periodical/zhgzwkssxdzzz201406013 [12] Zhou LN, Zhang JW, Wang JC, et al. Bone marrow stromal and Schwann cells from adult rats can interact synergistically to aid in peripheral nerve repair even without intercellular contact in vitro[J]. J Tissue Eng Regen Med, 2012, 6(7):579-588. doi: 10.1002/term.v6.7 [13] 尹东亮, 孙翀, 朱焕斌, 等.CTLA4-Ig基因修饰骨髓间充质干细胞抑制大鼠肝移植排斥反应[J].器官移植, 2014, 5(4):231-236. http://www.organtranspl.com/browse/detail/qkid/90/id/210.htmlYin DL, Sun C, Zhu HB, et al. Bone marrow mesenchymal stem cell modified by CTLA4-Ig gene can inhibit the rejection of liver transplantation in rats[J]. Organ Transplant, 2014, 5(4):231-236. http://www.organtranspl.com/browse/detail/qkid/90/id/210.html [14] 王彩霞, 张玉平, 陈小卫, 等.骨髓间充质干细胞减轻体外扩增的脐血造血干细胞衰老的作用[J].中华器官移植杂志, 2013, 34(7):432-435. http://d.wanfangdata.com.cn/Periodical/zhqgyz98201307013Wang CX, Zhang YP, Chen XW, et al. The protection of senescence by bone marrow mesenchymal stem cells in human cord blood CD34+ expanded cells[J]. Chin J Organ Transplant, 2013, 34(7):432-435. http://d.wanfangdata.com.cn/Periodical/zhqgyz98201307013 [15] Catacchio I, Berardi S, Reale A, et al. Evidence for bone marrow adult stem cell plasticity:properties, molecular mechanisms, negative aspects, and clinical applications of hematopoietic and mesenchymal stem cells transdifferentiation[J]. Stem Cells Int, 2013:589139. https://www.hindawi.com/journals/sci/2013/589139/ [16] 刘立宝, 黄蕾, 张军航, 等.Caspase-3 siRNA减少骨髓间充质干细胞稳定株细胞凋亡的实验研究[J].器官移植, 2014, 5(5):283-288. http://www.organtranspl.com/browse/detail/qkid/82/id/190.htmlLiu LB, Huang L, Zhang JH, et al.Experimental study on the Caspase-3 siRNA decreases apoptosis in stable cell lines of bone marrow mesenchymal stem cells[J]. Organ Transplant, 2014, 5(5):283-288. http://www.organtranspl.com/browse/detail/qkid/82/id/190.html [17] 陈规划, 杨扬, 张琪, 等.间充质干细胞应用现状及展望[J/CD].中华肝脏外科手术学电子杂志, 2013, 2(4):1-3. http://d.wanfangdata.com.cn/Periodical/zhgzwkssxdzzz201304001Chen GH, Yang Y, Zhang Q, et al. Application status and prospects of mesenchymal stem cells[J/CD]. Chin J Hepat Surg:Electr Edit, 2013, 2(4):1-3. http://d.wanfangdata.com.cn/Periodical/zhgzwkssxdzzz201304001 [18] 柴树宏, 鲍艳春.人脐带间充质干细胞与骨髓间充质干细胞分化为胰岛素分泌细胞的能力比较[J].实用医学杂志, 2014, 30(1):52-54. http://d.wanfangdata.com.cn/Periodical/syyxzz201401019Chai SH, Bao YC. Comparison of umbilical cord mesenchymal stem cells and bone marrow mesenchymal stem cells to differentiate into insulin-producing cells the ability[J]. J Pract Med, 2014, 30(1):52-54. http://d.wanfangdata.com.cn/Periodical/syyxzz201401019 [19] Hou SY, Zhang HY, Quan DP, et al. Tissue-engineered peripheral nerve grafting by differentiated bone marrow stromal cells[J]. Neuroscience, 2006, 140(1):101-110. doi: 10.1016/j.neuroscience.2006.01.066 -





 下载:
下载: