Effects of hepatocellular carcinoma-associated fibroblasts on differentiation of monocyte-derived dendritic cells
-
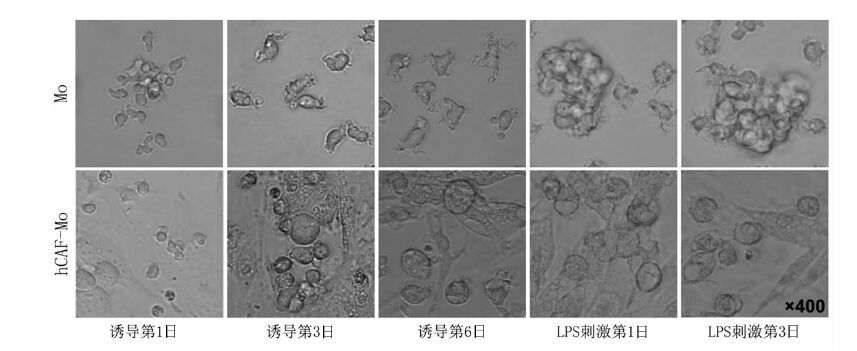
摘要: 目的 研究肝癌相关成纤维细胞(hCAF)在单核细胞(Mo)来源树突状细胞(DC)分化过程中所起到的作用。 方法 采用酶消化法从肝癌组织中分离培养获得人hCAF;采用密度梯度离心法获取健康人外周血单个核细胞(PBMC),经密度梯度离心及磁珠分离法从健康人外周血浓缩白细胞中获得CD14+Mo。hCAF-CD14+Mo(1:10)和CD14+Mo细胞在第1日及第4日均加入粒细胞-巨噬细胞集落刺激因子(20 ng/ml)和白细胞介素4(20 ng/ml),共诱导6 d。两组细胞分为两份,一半加入脂多糖(LPS)200 ng/ml刺激3 d,另一半不加入LPS作为对照。最后细胞共分为4组:hCAF-Mo组、hCAF-Mo+LPS组、不成熟DC(iDC)组及成熟DC(mDC)组。采用倒置荧光显微镜观察4组细胞的形态变化。各组细胞分为两部分,一部分细胞采用流式细胞术检测细胞表面分子CD83、CD80、CD1a的表达情况,另一部分细胞分别与预染CFSE的淋巴细胞共培养5 d,采用流式细胞术检测T淋巴细胞增殖情况。 结果 倒置显微镜结果显示在hCAF干扰下Mo不能分化为DC。CD83、CD1a、CD80的表达在iDC组分别为(3.2±0.7)%、(61.7±8.4)%、(30.1±0.9)%,mDC组分别为(80.1±2.8)%、(83.2±6.0)%、(96.1±1.9)%,hCAF-Mo组分别为(1.6±0.9)%、(1.8±0.9)%、(16.0±3.2)%,hCAF-Mo+LPS组分别为(9.0±1.2)%、(1.1±0.4)%、(58.4±3.6)%。hCAF-Mo组与iDC组的CD1a、CD80表达差异均有统计学意义(均为P<0.01)。iDC组的CD83表达极低,因此hCAF-Mo组与iDC组的CD83表达差异没有统计学意义(P>0.05)。hCAF-Mo+LPS组与mDC组的3种表型表达差异均有统计学意义(均为P<0.01)。iDC组的T淋巴细胞增殖率为(3.3±0.9)%,mDC组为(34.5±7.3)%,hCAF-Mo组为(5.3±1.2)%,hCAF-Mo+LPS组为(7.0±1.2)%。与iDC组相比,mDC组能有效地刺激T淋巴细胞增殖。hCAF-Mo组和hCAF-Mo+LPS组刺激T淋巴细胞增殖的能力均较弱,与mDC组相比差异有统计学意义(均为P<0.01)。 结论 hCAF可明显抑制Mo分化成DC,在肝癌的免疫逃逸过程中发挥重要的作用。Abstract: Objective To study the effects of hepatocellular carcinoma-associated fibroblasts(hCAF) on the differentiation of monocyte(Mo)-derived dendritic cell(DC). Methods The human hCAF were obtained from hepatic carcinoma tissue by enzyme digestion method. The healthy human peripheral blood mononuclear cells(PBMC) were prepared by density gradient centrifugation and CD14+Mo from healthy human peripheral white blood cells were purified by density gradient centrifugation and magnetic bead separation. hCAF-CD14+Mo(1:10) and CD14+Mo were treated by granulocyte-macrophage colony-stimulation factor(GM-CSF) (20 ng/ml) and interleukin(IL)-4 (20 ng/ml) which were added in day 1 and day 4 for 6 days. And then the cells of two groups were divided into two halves: one with or without the further stimulation of lipopolysaccharides(LPS, 200 ng/ml) for 3 days respectively. Finally the cells were divided into 4 groups: hCAF-Mo, hCAF-Mo+LPS, immature DC(iDC) and mature DC(mDC) group. Morphological changes of cells in 4 groups were observed by inverted fluorescence microscope. Cells in each group were divided into two parts. The expression of CD83,CD80, CD1a of cells in one part were detected by flow cytometry. And ability of T cells proliferation in the other part was analyzed by flow cytometry after co-culturing with CFSE-labeled T cells for 5 days. Results The result of inverted microscope showed that Mo couldn't differentiate into DC by the interference of hCAF. The expressions of CD83, CD1a and CD80 were (3.2±0.7)%,(61.7±8.4)%,(30.1±0.9)% respectively in iDC group,(80.2±2.8)%, (83.15±6.0)%,(96.1±1.9)% in mDC group,(1.6±0.9)%,(1.8±0.9)%,(16.0±3.2)% in hCAF-Mo group,(9.0±1.2)%、(1.1±0.4)%、(58.4±3.6)% in hCAF-Mo+LPS group. There was significant difference of the expressions of CD1a and CD80 between hCAF-Mo and iDC groups(both in P<0.01). Due to the extremely low expression of CD83 in iDC group, there was no significant difference between hCAF-Mo and iDC groups(P>0.05). There was significant difference of the expressions of CD83,CD1a and CD80 between hCAF-Mo+LPS and mDC groups (all in P<0.01). Proliferation rate of T cells were (3.3±0.9)% in iDC group, (34.5±7.3)% in mDC group,(5.3±1.2)% in hCAF-Mo group,(7.0±1.2)% in hCAF-Mo+LPS group. Compared with iDC group, mDC could promote the proliferation of T cells effectively. And the proliferation of T cells in hCAF-Mo and hCAF-Mo+LPS group was lower than that in mDC group(P<0.01). Conclusions hCAF can suppress the differentiation of Mo-derived DC, which may play an important role in immune escape of hepatocellular carcinoma.
-
-
[1] Mazzocca A, Dituri F, Lupo L,et al.Tumor-secreted lysophostatidic acid accelerates hepatocellular carcinoma progression by promoting differentiation of peritumoral fibroblasts in myofibroblasts[J]. Hepatology, 2011,54(3):920-930. doi: 10.1002/hep.24485 [2] Jia CC, Wang TT, Liu W, et al. Cancer-associated fibroblasts from hepatocellular carcinoma promote malignant cell proliferation by HGF secretion[J]. PLoS One, 2013, 8(5):e63243. doi: 10.1371/journal.pone.0063243 [3] 汪田甜,贾昌昌,张琪,等. 肝癌相关成纤维细胞的培养和鉴定及其对肝癌细胞增殖的影响[J]. 吉林大学学报, 2012, 38(3):438-442. http://www.cnki.com.cn/Article/CJFDTOTAL-BQEB201203014.htmWang TT, Jia CC, Zhang Q,et al. Culture and identification of hepatocellular carcinoma associated fibroblasts and their effects on proliferation of hepatocellular carcinoma cells[J]. J Jilin Univ: Med Ed, 2012, 38(3):438-442. http://www.cnki.com.cn/Article/CJFDTOTAL-BQEB201203014.htm [4] Mantovani A, Allavena P, Sica A,et al.Cancer-related inflammation[J].Nature, 2008, 454(7203):436-444. doi: 10.1038/nature07205 [5] Hembruff SL, Jokar I, Yang L, et al. Loss of transforming growth factor-beta signaling in mammary fibroblasts enhances CCL2 secretion to promote mammary tumor progression through macrophage-dependent and -independent mechanisms[J]. Neoplasia, 2010, 12(5):425-433. doi: 10.1593/neo.10200 [6] Augsten M, Hägglöf C, Olsson E,et al.CXCL14 is an autocrine growth factor for fibroblasts and acts as a multi-modal stimulator of prostate tumor growth[J]. Proc Natl Acad Sci U S A, 2009, 106(9):3414-3419. doi: 10.1073/pnas.0813144106 [7] Kobayashi N, Miyoshi S, Mikami T,et al.Hyaluronan deficiency in tumor stroma impairs macrophage trafficking and tumor neovascularization[J]. Cancer Res, 2010,70(18):7073-7083. doi: 10.1158/0008-5472.CAN-09-4687 [8] Li T, Yang Y, Hua X,et al. Hepatocellular carcinoma-associated fibroblasts trigger NK cell dysfunction via PGE2 and IDO[J]. Cancer Lett, 2012, 318(2):154-161. doi: 10.1016/j.canlet.2011.12.020 [9] Su X, Ye J, Hsueh EC,et al.Tumor microenvironments direct the recruitment and expansion of human Th17 cells[J]. J Immunol, 2010, 184(3):1630-1641. doi: 10.4049/jimmunol.0902813 [10] Nazareth MR, Broderick L, Simpson-Abelson MR,et al. Characterization of human lung tumor-associated fibroblasts and their ability to modulate the activation of tumor-associated T cells[J]. J Immunol,2007,178(9):5552-5562. doi: 10.4049/jimmunol.178.9.5552 [11] Barnas JL, Simpson-Abelson MR, Brooks SP, et al. Reciprocal functional modulation of the activation of T lymphocytes and fibroblasts derived from human solid tumors[J]. J Immunol, 2010, 185(5):2681-2692. doi: 10.4049/jimmunol.1000896 [12] Yang L, Pang Y, Moses HL. TGF-beta and immune cells: an important regulatory axis in the tumor microenvironment and progression[J]. Trends Immunol,2010, 31(6):220-227. doi: 10.1016/j.it.2010.04.002 [13] van de Ven R, Lindenberg JJ, Oosterhoff D, et al. Dendritic cell plasticity in tumor-conditioned skin: CD14+ cells at the cross-roads of immune activation and suppression[J]. Front Immunol,2013,4:403. [14] 马麟麟.免疫抑制剂对树突状细胞的影响[J].器官移植,2013,4(6):370-372. http://www.organtranspl.com/browse/detail/qkid/0/id/255.htmlMa LL. Effect of immunosuppressive agents on the dendritic cell[J]. Organ Transplant,2013,4(6):370-372. http://www.organtranspl.com/browse/detail/qkid/0/id/255.html [15] Mailliard RB, Dallal RM, Son YI,et al.Dendritic cells promote T-cell survival or death depending upon their maturation state and presentation of antigen[J].Immunol Invest,2000, 29(2):177-185. doi: 10.3109/08820130009062302 [16] 张海明,郑虹,邓永林,等.肝癌复发与未复发患者再次肝移植的预后分析[J].中华器官移植杂志,2013,34(7):407-410.Zhang HM, Zheng H, Deng YL, et al. Retransplantation of HCC patients with or without HCC recurrence[J]. Chin Organ Transplant,2013,34(7):407-410. [17] 陈规划, 张琪. 干细胞在肝移植中的应用现状及展望[J/CD]. 中华肝脏外科手术学电子杂志, 2012, 1(2): 70-73.Chen GH, Zhang Q. Stem cell therapy in the liver transplantation[J/CD].Chin J Hapat Surg(Electronic Edition), 2012, 1(2): 70-73. [18] Reinhard G, Märten A, Kiske SM,et al.Generation of dendritic cell-based vaccines for cancer therapy[J]. Br J Cancer,2002, 86(10):1529-1533. doi: 10.1038/sj.bjc.6600316 -





 下载:
下载: