Expression of BZW2 and IVD in hepatocellular carcinoma tissues and effect on prognosis of liver transplant recipients with hepatocellular carcinoma
-
摘要:目的 探讨碱性亮氨酸拉链和W2结构域2(BZW2)及异戊酰辅酶A脱氢酶(IVD)在肝细胞癌(肝癌)中的表达及其对肝移植受者预后的影响。方法 回顾性分析87例肝癌肝移植受者的病理标本及临床资料,分析肝癌肝移植受者术后肿瘤复发转移情况。采用免疫组织化学染色检测BZW2和IVD的表达情况,分析BZW2和IVD与肝癌临床病理参数的关系及其对肝移植术后肿瘤复发和受者预后的影响。结果 87例受者中,共有31例肿瘤复发,复发率为36%,复发时间为术后2~49个月,复发时间中位数为7个月。免疫组织化学染色结果显示,肝癌组织的BZW2阳性表达率高于正常肝组织(76%比30%),IVD阳性表达率低于正常肝组织(51%比69%),差异均有统计学意义(均为P < 0.01)。BZW2表达与肿瘤直径、有否肿瘤包膜有关(均为P < 0.05),IVD表达与肿瘤直径,甲胎蛋白(AFP)水平,肿瘤、淋巴结、转移(TNM)分期,有否血管侵犯有关(均为P < 0.05)。BZW2高表达组受者肿瘤累积复发率高于BZW2低表达组,累积生存率低于BZW2低表达组;IVD低表达组患者肿瘤累积复发率高于IVD高表达组,累积生存率低于IVD高表达组(均为P < 0.05)。结论 在肝癌组织中,BZW2蛋白表达水平上调,IVD蛋白表达水平下调。且BZW2高表达和IVD低表达的肝移植受者的累积复发率较高,累积生存率较低。
-
关键词:
- 肝细胞癌 /
- 肝移植 /
- 碱性亮氨酸拉链和W2结构域2(BZW2) /
- 异戊酰辅酶A脱氢酶(IVD) /
- 甲胎蛋白 /
- 血管侵犯 /
- 肿瘤包膜 /
- 肿瘤分期 /
- 肿瘤复发
Abstract:Objective To investigate the expression levels of basic leucine zipper and W2 domain 2 (BZW2) and isovaleryl-CoA dehydrogenase (IVD) in hepatocellular carcinoma (HCC) and evaluate their effect on clinical prognosis of liver transplant recipients with HCC.Methods Pathological specimens and clinical data of 87 liver transplant recipients with HCC were collected and retrospectively analyzed. The recurrence and metastasis of HCC after liver transplantation were assessed. Immunohistochemical staining was used to detect the expression levels of BZW2 and IVD. The relationship between BZW2, IVD and clinicopathological parameters of HCC and their effect on postoperative recurrence and clinical prognosis of the recipients was analyzed.Results Among 87 recipients, 31 cases recurred with a recurrence rate of 36%. HCC recurred at postoperative 2-49 months and the median recurrence time was postoperative 7 months. Immunohistochemical staining demonstrated that the positive expression rate of BZW2 in the HCC tissues was significantly higher than that in normal liver tissues (76% vs. 30%), and the positive expression rate of IVD was significantly lower compared with that in normal liver tissues (51% vs. 69%) (both P < 0.01). BZW2 expression was significantly correlated with tumor diameter and tumor capsule (both P < 0.05), whereas IVD expression was significantly associated with tumor diameter, alpha-fetoprotein (AFP) level, tumor, node and metastasis (TNM) staging and whether vascular invasion was found or not (all P < 0.05). In the high BZW2 expression group, the cumulative recurrence rate of HCC was significantly higher and the cumulative survival rate was significantly lower than those in the low BZW2 expression group. In the low IVD expression group, the cumulative recurrence rate of HCC was significantly higher and the cumulative survival rate was significantly lower compared with those in the high IVD expression group (all P < 0.05).Conclusions The expression level of BZW2 protein is up-regulated, whereas that of IVD protein is down-regulated in the HCC tissues. Moreover, the cumulative recurrence rate of HCC is relatively high and the cumulative survival rate is relatively low in liver transplant recipients with high BZW2 expression and low IVD expression. -
原发性肝癌是我国第4位常见恶性肿瘤及第2位肿瘤致死病因,肝细胞癌(肝癌)是其主要病理类型,约占90%[1-2]。肝移植是根治性治疗肝癌的方法之一,但肝移植术后5年肿瘤复发率达20.0%~57.8%,复发受者的中位生存期仅为7~16个月[3-4]。既往研究表明,肝癌肝移植术后复发与肿瘤分期、血管侵犯、免疫抑制剂用量等因素相关[5-8],但仍缺乏有效的基因标志物。
碱性亮氨酸拉链和W2结构域2(basic leucine zipper and W2 domain 2,BZW2)是碱性亮氨酸拉链(basic leucine zipper,bZIP)转录因子超家族成员之一,是一种进化高度保守的蛋白,可通过黏附蛋白参与细胞间的黏附[9-10]。既往研究表明,BZW2在膀胱癌、骨肉瘤、结肠癌、喉乳头状瘤中表达上调且与预后相关[11-14]。异戊酰辅酶A脱氢酶(isovaleryl-CoA dehydrogenase,IVD)催化异戊基辅酶A转化为3-甲基丙烯酰辅酶A,其表达异常可影响亮氨酸代谢并导致异戊酸血症[15-18]。然而,笔者查阅文献,尚未见BZW2和IVD对肝癌肝移植术后的肿瘤复发和预后影响的研究报道。本研究旨在探讨肝癌组织中BZW2和IVD的表达及其与肝癌肝移植术后肿瘤复发和预后的关系。
1. 资料与方法
1.1 一般资料
回顾性分析87例于2015年1月至2018年12月在中国人民解放军联勤保障部队第九〇〇医院肝胆外科行同种异体原位肝移植受者的临床资料及病理标本,包括肝癌组织和正常肝组织。其中男76例,女11例,年龄20~73岁。纳入标准:(1)术后病理确诊为肝细胞癌;(2)术前病历资料及术后随访资料完整;(3)病理标本保存齐全;(4)符合美国加州大学旧金山分校(University of California, San Francisco,UCSF)标准。排除标准:(1)围手术期死亡;(2)合并严重基础疾病;(3)失访;(4)非肿瘤相关性死亡。
所有受者通过门诊复查、住院治疗及电话进行随访,术后2年内每3个月复查1次,术后2年后每半年复查1次。随访至术后60个月或受者死亡。随访内容包括血常规、肝功能、甲胎蛋白(alpha-fetoprotein,AFP)水平及影像学检查等。
供者来源均为心脏死亡器官捐献(donation after cardiac death,DCD)。术后免疫抑制方案为糖皮质激素+他克莫司+吗替麦考酚酯。抗乙型病毒性肝炎(乙肝)复发方案为恩替卡韦+乙肝人免疫球蛋白。本研究经中国人民解放军联勤保障部队第九〇〇医院伦理委员会批准。
1.2 实验方法
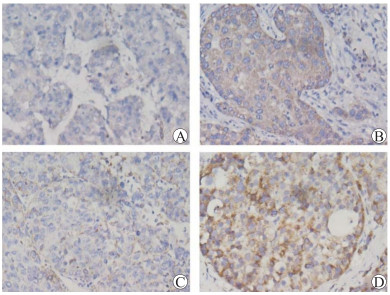
采用免疫组织化学染色检测BZW2和IVD的表达。肝组织石蜡切片经二甲苯脱蜡,乙醇梯度洗涤后进行抗原修复。3%过氧化氢溶液孵育10 min,二抗来源羊血清封闭液室温孵育30 min。滴加抗BZW2抗体(1∶200)、抗IVD抗体(1∶200)(抗体均购自英国Abcam公司),4 ℃孵育过夜。次日37 ℃复温,滴加增强剂,室温孵育20 min后,滴加100 μL增强酶标山羊抗兔IgG聚合物,室温孵育20 min。3,3'-二氨基联苯胺(3, 3'-diaminobenzidine,DAB)染色1 min,显微镜下观察颜色反应,自来水冲洗10 min,苏木素复染40 s,再用自来水冲洗10 min,烘干后中性树脂封片。在显微镜下观察染色情况,BZW2和IVD在细胞质中呈现淡黄、棕黄或褐色颗粒。根据阳性细胞着色强度和阳性细胞比例进行评分:无染色为0分,淡黄色为1分,棕黄色为2分,褐色为3分;阳性细胞数 < 10%为0分,10%~25%为1分,26%~50%为2分, > 50%为3分。两项得分相乘,≤4分为低表达, > 4分为高表达。
1.3 研究内容
分析肝癌肝移植受者术后肿瘤复发转移情况。检测肝癌组织和正常肝脏组织中BZW2和IVD的表达情况,分析肝癌肝移植受者术前原发肿瘤病灶BZW2和IVD的表达与临床病理参数[年龄,性别,肿瘤直径,AFP水平,肿瘤、淋巴结、转移(tumor,node,metastasis,TNM)分期[19],有否血管侵犯,有否肿瘤包膜,肿瘤数量]之间的关系;分析肝癌肝移植受者BZW2和IVD的表达情况与术后复发及生存情况的关系。
1.4 统计学方法
采用SPSS 23.0软件进行统计学分析。计数资料以率表示,比较采用χ2检验或Fisher确切概率检验。采用Kaplan-Meier绘制生存曲线,采用log-rank检验进行生存分析。P < 0.05为差异有统计学意义。
2. 结果
2.1 肝癌肝移植受者术后肿瘤复发转移情况
截止至2021年3月,87例肝癌肝移植受者随访时间3~68个月,中位随访时间33个月。随访期间共有31例肿瘤复发,复发率为36%,复发时间为术后2~49个月,复发时间中位数为7个月。复发转移部位包括肺20例(65%)、肝11例(35%)、骨9例(29%)、腹腔3例(10%)、肾上腺l例(3%)。
2.2 BZW2和IVD在肝癌组织和肝组织中的表达情况
免疫组织化学染色结果显示,阳性染色部位主要在细胞浆(图 1)。肝癌组织的BZW2阳性表达率为76%(66/87),高于正常肝组织的30%(26/87),差异有统计学意义(χ2=26.903,P < 0.01);肝癌组织的IVD阳性表达率为51%(44/87),低于正常肝组织的69%(60/87),差异有统计学意义(χ2=13.167,P < 0.01)。
2.3 BZW2和IVD与肝癌临床病理参数的关系
BZW2、IVD与肝癌临床病理参数关系的分析结果见表 1。BZW2的表达水平与肿瘤直径、有否肿瘤包膜有关(均为P < 0.05),与其他临床病理参数无关(均为P > 0.05)。IVD的表达水平与肿瘤直径、AFP水平、TNM分期、有否血管侵犯有关(均为P < 0.05),与其他临床病理参数无关(均为P > 0.05)。
表 1 BZW2和IVD与肝癌肝移植临床病理参数的关系Table 1. Relationship between BZW2 and IVD and clinicopathological parameters of liver transplantation for hepatocellular carcinoma [n (%)]项目 n BZW2高表达 P值 IVD高表达 P值 年龄 0.96 0.46 ≤50岁 46 35(76) 25(54) > 50岁 41 31(76) 19(46) 性别 0.07 0.35 男 76 60(79) 40(53) 女 11 6(55) 4(36) 肿瘤直径 0.03 < 0.01 ≤5 cm 53 36(68) 33(62) > 5 cm 34 30(88) 11(32) AFP水平 0.68 < 0.01 ≤400 μg/L 59 44(75) 37(63) > 400 μg/L 28 22(79) 7(25) TNM分期 0.86 < 0.01 Ⅰ~Ⅱ期 65 49(75) 37(57) Ⅲ~Ⅳ期 22 17(77) 7(32) 血管侵犯 0.38 < 0.01 有 21 18(86) 4(19) 无 66 48(73) 40(61) 肿瘤包膜 < 0.01 0.10 有 18 8(44) 6(33) 无 69 58(84) 38(55) 肿瘤数量 0.08 0.19 单发 59 48(81) 27(46) 多发 28 18(64) 17(61) 2.4 BZW2和IVD表达与肝癌肝移植受者术后复发和生存的关系
87例肝癌肝移植受者术后1、3、5年累积复发率分别为27.6%、36.9%、40.2%;l、3、5年累积生存率分别为80.5%、47.7%、34.7%。各组肝移植受者的生存曲线见图 2,BZW2高表达组受者肿瘤累积复发率高于BZW2低表达组(χ2=4.938,P < 0.05),累积生存率低于BZW2低表达组(χ2=4.464,P < 0.05),IVD低表达组受者肿瘤累积复发率高于IVD高表达组(χ2=5.606,P < 0.05),累积生存率低于IVD高表达组(χ2=5.703,P < 0.05)。
3. 讨论
肝移植是肝癌的根治性治疗手段之一,肿瘤复发是肝癌肝移植术后面临的主要问题[4, 20-25]。肝癌肝移植术后有无肿瘤复发则是受者术后能否长期生存的关键,但目前仍缺乏有效评估预测肝癌肝移植术后肿瘤复发和转移的方法。
BZW2是一种进化上高度保守的蛋白,与翻译起始因子eIF2和eIF3相互作用,进而调节蛋白质合成[26]。Huang等[13]认为,BZW2在大多数实体肿瘤中作为一种致癌基因,涉及多种信号通路,他们发现BZW2可通过激活细胞外信号调节激酶(extracellular signal-regulated kinase,ERK)/丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)信号通路,促进结直肠癌的细胞增殖、侵袭和迁移。Jin等[27]研究表明,BZW2过表达显著激活肝癌细胞的磷脂酰肌醇-3-激酶(phosphatidylinositol-3-kinase,PI3K)/蛋白激酶B(protein kinase B,Akt)/哺乳动物雷帕霉素靶蛋白(mammalian target of rapamycin,mTOR)信号通路,促进肿瘤细胞增殖,同时降低PI3K抑制剂的抑制效率,导致肿瘤发生和进展。而下调BZW2的表达,可以导致G1期细胞周期停滞[11],并通过抑制Akt/mTOR信号通路,进而抑制骨肉瘤细胞的生长[12]。近年来,也有关于BZW2表达在肝癌细胞侵袭转移过程中的研究[26, 28-29],但缺乏临床资料验证且未应用于肝移植术后复发的预测中。IVD催化异戊基辅酶A转化为3-甲基丙烯酰辅酶A,既往研究表明其表达与某些微生物能量代谢相关[30-32],在人体中IVD的表达异常可影响亮氨酸代谢并导致异戊酸血症,Lee等[33]发现肝癌门静脉血栓形成中代谢蛋白水平下调,但未进行更深入研究。
本研究结果表明BZW2的表达在肝癌组织中上调,IVD的表达在肝癌组织中下调,且BZW2高表达与肝癌直径较大、无肿瘤包膜有关,IVD低表达与肿瘤直径较大、AFP水平较高、肿瘤TNM分期Ⅲ~Ⅳ期、有血管侵犯有关。生存分析结果显示BZW2高表达组和IVD低表达组受者肿瘤复发率较高,生存率较低,提示肝癌组织BZW2高表达和IVD低表达的受者可能存在肿瘤复发转移的潜在风险。
综上所述,BZW2和IVD有可能成为预测肝癌肝移植受者预后的潜在指标。但由于本研究样本量较小,且为单中心回顾性研究,上述研究结果仍需进一步证实。后续研究中将对供肝中BZW2和IVD的表达情况进行检测,排除相关干扰。关于BZW2和IVD如何调控肿瘤增殖和转移,也有待进一步深入研究。
-
表 1 BZW2和IVD与肝癌肝移植临床病理参数的关系
Table 1 Relationship between BZW2 and IVD and clinicopathological parameters of liver transplantation for hepatocellular carcinoma [n (%)]
项目 n BZW2高表达 P值 IVD高表达 P值 年龄 0.96 0.46 ≤50岁 46 35(76) 25(54) > 50岁 41 31(76) 19(46) 性别 0.07 0.35 男 76 60(79) 40(53) 女 11 6(55) 4(36) 肿瘤直径 0.03 < 0.01 ≤5 cm 53 36(68) 33(62) > 5 cm 34 30(88) 11(32) AFP水平 0.68 < 0.01 ≤400 μg/L 59 44(75) 37(63) > 400 μg/L 28 22(79) 7(25) TNM分期 0.86 < 0.01 Ⅰ~Ⅱ期 65 49(75) 37(57) Ⅲ~Ⅳ期 22 17(77) 7(32) 血管侵犯 0.38 < 0.01 有 21 18(86) 4(19) 无 66 48(73) 40(61) 肿瘤包膜 < 0.01 0.10 有 18 8(44) 6(33) 无 69 58(84) 38(55) 肿瘤数量 0.08 0.19 单发 59 48(81) 27(46) 多发 28 18(64) 17(61) -
[1] ZHENG R, QU C, ZHANG S, et al. Liver cancer incidence and mortality in China: temporal trends and projections to 2030[J]. Chin J Cancer Res, 2018, 30(6)571-579. DOI: 10.21147j.issn.1000-9604.2018.06.01.
[2] SIEGEL RL, MILLER KD, JEMAL A. Cancer statistics, 2020[J]. CA Cancer J Clin, 2020, 70(1): 7-30. DOI: 10.3322/caac.21590.
[3] FILGUEIRA NA. Hepatocellular carcinoma recurrence after liver transplantation: risk factors, screening and clinical presentation[J]. World J Hepatol, 2019, 11(3): 261-272. DOI: 10.4254/wjh.v11.i3.261.
[4] SPOSITO C, CUCCHETTI A, MAZZAFERRO V. Assessing competing risks for death following liver transplantation for hepatocellular carcinoma[J]. Dig Dis Sci, 2019, 64(4): 1001-1007. DOI: 10.1007/s10620-019-05538-1.
[5] 王建丰, 曾凯宁, 李海波, 等. 微血管侵犯对肝癌肝移植受者预后影响的临床研究[J]. 器官移植, 2021, 12(3): 309-316. DOI: 10.3969/j.issn.1674-7445.2021.03.009. WANG JF, ZENG KN, LI HB, et al. Clinical study of microvascular invasion on prognosis of recipients after liver transplantation for liver cancer[J]. Organ Transplant, 2021, 12(3): 309-316. DOI: 10.3969/j.issn.1674-7445.2021.03.009.
[6] AL-AMERI AAM, WEI X, WEN X, et al. Systematic review: risk prediction models for recurrence of hepatocellular carcinoma after liver transplantation[J]. Transpl Int, 2020, 33(7): 697-712. DOI: 10.1111/tri.13585.
[7] WANG F, BRESLIN SJP, QIU W. Novel oncogenes and tumor suppressor genes in hepatocellular carcinoma[J]. Liver Res, 2021, 5(4): 195-203. DOI: 10.1016/j.livres.2021.06.001.
[8] NATU A, SINGH A, GUPTA S. Hepatocellular carcinoma: understanding molecular mechanisms for defining potential clinical modalities[J]. World J Hepatol, 2021, 13(11): 1568-1583. DOI: 10.4254/wjh.v13.i11.1568.
[9] CHOI W, CZERNIAK B, OCHOA A, et al. Intrinsic basal and luminal subtypes of muscle-invasive bladder cancer[J]. Nat Rev Urol, 2014, 11(7): 400-410. DOI: 10.1038/nrurol.2014.129.
[10] SOLOWAY MS. Bladder cancer: lack of progress in bladder cancer--what are the obstacles?[J]. Nat Rev Urol, 2013, 10(1): 5-6. DOI: 10.1038/nrurol.2012.219.
[11] GAO H, YU G, ZHANG X, et al. BZW2 gene knockdown induces cell growth inhibition, G1 arrest and apoptosis in muscle-invasive bladder cancers: a microarray pathway analysis[J]. J Cell Mol Med, 2019, 23(6): 3905-3915. DOI: 10.1111/jcmm.14266.
[12] CHENG DD, LI SJ, ZHU B, et al. Downregulation of BZW2 inhibits osteosarcoma cell growth by inactivating the Akt/mTOR signaling pathway[J]. Oncol Rep, 2017, 38(4): 2116-2122. DOI: 10.3892/or.2017.5890.
[13] HUANG L, CHEN S, FAN H, et al. BZW2 promotes the malignant progression of colorectal cancer via activating the ERK/MAPK pathway[J]. J Cell Physiol, 2020, 235(5): 4834-4842. DOI: 10.1002/jcp.29361.
[14] LIU J, YANG T, ZHANG Y, et al. Promotion of BZW2 by LINC00174 through miR-4500 inhibition enhances proliferation and apoptosis evasion in laryngeal papilloma[J]. Cancer Cell Int, 2020, 20: 471. DOI: 10.1186/s12935-020-01559-3.
[15] MOHSEN AW, VOCKLEY J. Kinetic and spectral properties of isovaleryl-CoA dehydrogenase and interaction with ligands[J]. Biochimie, 2015, 108: 108-119. DOI: 10.1016/j.biochi.2014.11.007.
[16] HYMAN DB, TANAKA K. Isovaleryl-CoA dehydrogenase activity in isovaleric acidemia fibroblasts using an improved tritium release assay[J]. Pediatr Res, 1986, 20(1): 59-61. DOI: 10.1203/00006450-198601000-00017.
[17] D'ANNIBALE OM, KOPPES EA, ALODAIB AN, et al. Characterization of variants of uncertain significance in isovaleryl-CoA dehydrogenase identified through newborn screening: an approach for faster analysis[J]. Mol Genet Metab, 2021, 134(1/2): 29-36. DOI: 10.1016/j.ymgme.2021.08.012.
[18] LI Y, ZHENG X, ZHU M, et al. MoIVD-mediated leucine catabolism is required for vegetative growth, conidiation and full virulence of the rice blast fungus magnaporthe oryzae[J]. Front Microbiol, 2019, 10: 444. DOI: 10.3389/fmicb.2019.00444.
[19] 中华人民共和国国家卫生健康委员会医政医管局. 原发性肝癌诊疗规范(2019年版)[J]. 临床肝胆病杂志, 2020, 36(2): 277-292. DOI: 10.3969/j.issn.1001-5256.2020.02.007. Chinese Journal of Clinical Bureau of Medical Administration, National Health Commission of the People's Republic of China. Guidelines for diagnosis and treatment of primary liver cancer in China (2019 edition)[J]. J Clin Hepatol, 2020, 36(2): 277-292. DOI: 10.3969/j.issn.1001-5256.2020.02.007.
[20] 汪国营. 肝癌肝移植相关热点问题探讨[J]. 中山大学学报(医学科学版), 2021, 42(1): 17-23. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSYK202101003.htm WANG GY. Hot issues related to liver transplantation for hepatocellular carcinoma[J]. J Sun Yat-sen Univ (Med Sci), 2021, 42(1): 17-23. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSYK202101003.htm
[21] 霍枫, 季茹, 陈建雄. 肝细胞癌肝移植供肝选择策略[J]. 中华消化外科杂志, 2020, 19(2): 135-138. DOI: 10.3760/cma.j.issn.1673-9752.2020.02.005. HUO F, JI R, CHEN JX. Selection of donor liver in liver transplantation for hepatocellular carcinoma[J]. Chin J Dig Surg, 2020, 19(2): 135-138. DOI: 10.3760/cma.j.issn.1673-9752.2020.02.005.
[22] VERNA EC, PATEL YA, AGGARWAL A, et al. Liver transplantation for hepatocellular carcinoma: management after the transplant[J]. Am J Transplant. 2020, 20(2): 333-347. DOI: 10.1111/ajt.15697.
[23] HOFFMAN D, MEHTA N. Recurrence of hepatocellular carcinoma following liver transplantation[J]. Expert Rev Gastroenterol Hepatol, 2021, 15(1): 91-102. DOI: 10.1080/17474124.2021.1823213.
[24] SAMUEL D, COILLY A. Management of patients with liver diseases on the waiting list for transplantation: a major impact to the success of liver transplantation[J]. BMC Med, 2018, 16(1): 113. DOI: 10.1186/s12916-018-1110-y.
[25] 刘召波, 栗光明. 肝移植术后肝癌复发防治进展[J]. 临床肝胆病杂志, 2021, 37(2): 267-271. DOI: 10.3969/j.issn.1001-5256.2021.02.006. LIU ZB, LI GM. Advances in the prevention and treatment of hepatocellular carcinoma recurrence after liver transplantation[J]. J Clin Hepatol, 2021, 37(2): 267-271. DOI: 10.3969/j.issn.1001-5256.2021.02.006.
[26] SINGH CR, WATANABE R, ZHOU D, et al. Mechanisms of translational regulation by a human eIF5-mimic protein[J]. Nucleic Acids Res, 2011, 39(19): 8314-8328. DOI: 10.1093/nar/gkr339.
[27] JIN X, LIAO M, ZHANG L, et al. Role of the novel gene BZW2 in the development of hepatocellular carcinoma[J]. J Cell Physiol, 2019, 234(9): 16592-16600. DOI: 10.1002/jcp.28331.
[28] LI G, LU A, CHEN A, et al. BZW2/5MP1 acts as a promising target in hepatocellular carcinoma[J]. J Cancer, 2021, 12(17): 5125-5135. DOI: 10.7150/jca.53282.
[29] LIU L, ZHAO J, PENG Y, et al. MiR-let-7a-5p inhibits invasion and migration of hepatoma cells by regulating BZW2 expression[J]. Onco Targets Ther, 2020, 13: 12269-12279. DOI: 10.2147/OTT.S278954.
[30] HIROTA T, IZUMI M, WADA S, et al. Vacuolar protein degradation via autophagy provides substrates to amino acid catabolic pathways as an adaptive response to sugar starvation in Arabidopsis thaliana[J]. Plant Cell Physiol, 2018, 59(7): 1363-1376. DOI: 10.1093/pcp/pcy005.
[31] SURGER MJ, ANGELOV A, STIER P, et al. Impact of branched-chain amino acid catabolism on fatty acid and alkene biosynthesis in micrococcus luteus[J]. Front Microbiol, 2018, 9: 374. DOI: 10.3389/fmicb.2018.00374.
[32] MILLERIOUX Y, MAZET M, BOUYSSOU G, et al. De novo biosynthesis of sterols and fatty acids in the Trypanosoma brucei procyclic form: carbon source preferences and metabolic flux redistributions[J]. PLoS Pathog, 2018, 14(5): e1007116. DOI: 10.1371/journal.ppat.1007116.
[33] LEE WC, CHOU HS, WU TJ, et al. Down-regulation of metabolic proteins in hepatocellular carcinoma with portal vein thrombosis[J]. Clin Proteomics, 2017, 14: 29. DOI: 10.1186/s12014-017-9164-y.
-
期刊类型引用(4)
1. 李玉杰,刘东璞. BZW2在恶性肿瘤中的研究进展. 医学理论与实践. 2024(22): 3811-3813 .  百度学术
百度学术
2. 李航. IVD行业微升级高精度灌装机设计研究. 机械管理开发. 2024(12): 96-98 .  百度学术
百度学术
3. 付雪,宁登,陈劲,杜鹏程,刘秋梦,姜立. eEF1A1通过HGF/c-MET通路调控肝癌细胞的迁移和侵袭能力. 中国组织化学与细胞化学杂志. 2023(02): 119-127 .  百度学术
百度学术
4. 袁泽南,蔡建业,李海波,余振宇,朱曙光,张剑文. 介入联合免疫及抗血管生成靶向一线转化治疗晚期肝癌的临床疗效. 中华消化外科杂志. 2022(S1): 15-19 .  百度学术
百度学术
其他类型引用(0)



 下载:
下载:



